ARTICLE SUMMARY:
The evolution in device-based diabetes management began more than 50 years ago. Today, fueled by US healthcare costs approaching $330 billion and exponential global growth in incidence and prevalence, device technologies—powered by the promise of data and analytics—are helping to simplify the lives of diabetic patients and at the same time combat the rise in the cost of care. In this edition, we take a look at DexCom’s acquisition of TypeZero Technologies.
Before we highlight a recent market development in the treatment of diabetes - a problematic condition of epidemic proportions that accounts for one in four healthcare dollars spent in the US - let’s look back for a moment at how far we’ve come. 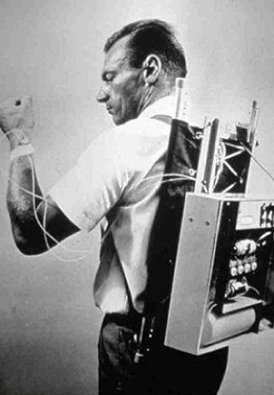
In 1963, the same year that President John F. Kennedy was assassinated, Martin Luther King, Jr. delivered his "I Have a Dream" speech, and Zoning Improvement Plan (ZIP) codes were implemented in the US, the first closed-loop insulin pump device for the treatment of type 1 diabetes (T1D) was invented. Designed by Dr. Arnold Kadish of Los Angeles, CA, the device worked by providing continuous insulin to the body together with automatic blood glucose sensing. However, due to its size - described as being similar to “an Army backpack” - Kadish’s device was deemed impractical for daily use (see image here).
Eleven years later, in 1974 - the year that Richard Nixon became the first US president to resign from office - the first bedside, computer-controlled closed-loop insulin pump, Biostator, was 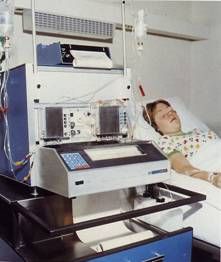 developed by Miles Laboratory Inc. (Elkhart, IN). The system, pictured below, was designed to simulate the function of normal pancreatic cells and consisted of: a pump which controlled continual withdrawal and mixing of blood; a glucose analyzer for continuous analysis of blood glucose concentration; a computer programmed with a set of algorithms to calculate the amount of insulin or dextrose to be infused based on blood glucose level; a computer-operated infusion pump for insulin or dextrose delivery; and a printer/plotter for minute-by-minute blood glucose recording.
developed by Miles Laboratory Inc. (Elkhart, IN). The system, pictured below, was designed to simulate the function of normal pancreatic cells and consisted of: a pump which controlled continual withdrawal and mixing of blood; a glucose analyzer for continuous analysis of blood glucose concentration; a computer programmed with a set of algorithms to calculate the amount of insulin or dextrose to be infused based on blood glucose level; a computer-operated infusion pump for insulin or dextrose delivery; and a printer/plotter for minute-by-minute blood glucose recording.
These early achievements marked the beginning of a competitive, five-decade-long race to develop a portable, closed-loop insulin pump and continuous glucose monitoring (CGM) system, also called an artificial pancreas system. (Our MedTech Strategist editorial team covers the rapidly evolving diabetes device space extensively; please refer to our archives.)
Recent device evolution has been sparked by exponential global growth in the incidence and prevalence, and costs, of diabetes - the seventh leading cause of death in the US as of 2015. As of the same year, 30.3 million Americans (9.4% of the population) had diabetes, with 23.1 million of that figure being diagnosed and 7.2 million undiagnosed, according to the American Diabetes Association. Approximately 1.25 million American children and adults have insulin-dependent T1D. The total cost of diagnosed diabetes in the US in 2017 was a staggering $327 billion – and that is a 26% increase just since 2012. Diabetes is a multi-edged sword, as it is associated with a host of costly complications including kidney disease, high blood pressure, diabetic ulcers, and stroke. There is an enormous demand for smart technologies to help simplify the lives of people with diabetes, and at the same time combat out-of-control healthcare costs.
In this week’s edition of “In MedTech History,” we highlight a recent development in the diabetes device space focused on delivering on these lofty goals, powered by data and analytics. DexCom Inc. has acquired TypeZero Technologies Inc., as part of DexCom’s goal of launching an integrated, automated insulin delivery system in 2019.
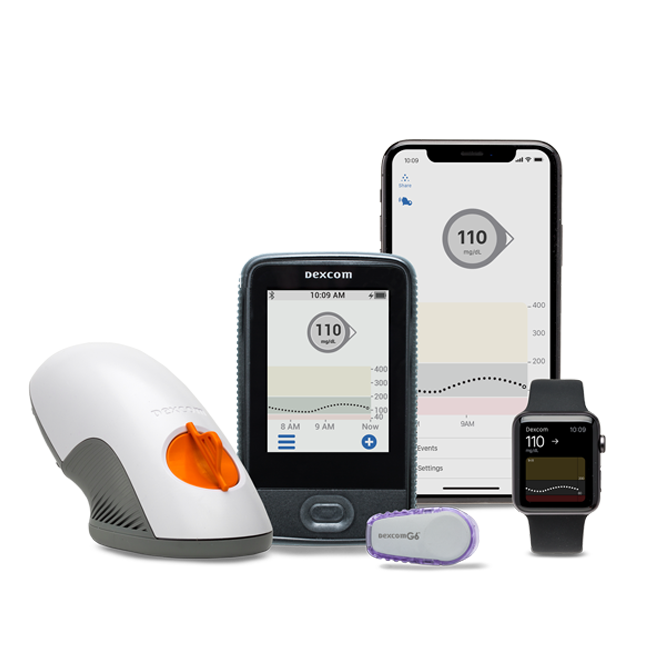
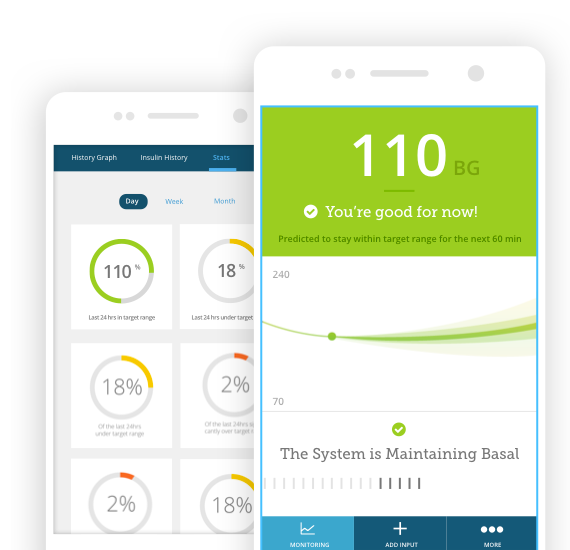 On August 22, CGM company DexCom Inc. announced that it had acquired five-year-old TypeZero Technologies Inc., for an undisclosed sum. TypeZero has developed a diabetes patient system and algorithm, inControl, which provides patients with a mobile app and insulin regulation system (see product image at right). DexCom’s zero-fingerstick G6 CGM System for people with diabetes ages two years and up was FDA-approved in March and CE mark-cleared in June (see image of the G6 product family below). (This follows FDA approval of Abbott Laboratories Inc.’s FreeStyle Libre last September, which also does not require any fingerstick calibrations and is approved for those 18 and over.)
On August 22, CGM company DexCom Inc. announced that it had acquired five-year-old TypeZero Technologies Inc., for an undisclosed sum. TypeZero has developed a diabetes patient system and algorithm, inControl, which provides patients with a mobile app and insulin regulation system (see product image at right). DexCom’s zero-fingerstick G6 CGM System for people with diabetes ages two years and up was FDA-approved in March and CE mark-cleared in June (see image of the G6 product family below). (This follows FDA approval of Abbott Laboratories Inc.’s FreeStyle Libre last September, which also does not require any fingerstick calibrations and is approved for those 18 and over.)
 “DexCom and TypeZero, along with the University of Virginia [from which TypeZero’s technology was licensed], have had a long and productive relationship in advancing technologies to improve diabetes management, and this transaction is a logical next step as the partnership makes progress,” a DexCom representative told the Community Blog. In addition, DexCom has a number of decision-support features in development to enhance its Clarity software and eventually patients’ real-time experience. TypeZero has been active in this area and will augment and accelerate DexCom’s roadmap, the DexCom representative told us.
“DexCom and TypeZero, along with the University of Virginia [from which TypeZero’s technology was licensed], have had a long and productive relationship in advancing technologies to improve diabetes management, and this transaction is a logical next step as the partnership makes progress,” a DexCom representative told the Community Blog. In addition, DexCom has a number of decision-support features in development to enhance its Clarity software and eventually patients’ real-time experience. TypeZero has been active in this area and will augment and accelerate DexCom’s roadmap, the DexCom representative told us.
The DexCom G6 is a patch device about the size of a quarter that is applied to the skin of the abdomen. It contains a small sensor that continuously measures the amount of glucose in body fluid. The device, which is replaced by the patient every 10 days, transmits real-time glucose readings every five minutes to a compatible display device such as a mobile medical app on a cell phone. It will trigger an alarm when a patient’s blood sugar enters a danger zone, soaring too high or dropping too low. When integrated with an automated insulin dosing system, a rise in blood sugar will trigger the release of insulin from the pump.
Notably, the FDA has recently paved the way for faster CGM innovation. In March, the agency announced that the DexCom G6 integrated continuous glucose monitoring (iCGM) system is the first type of CGM system approved to be used as part of an integrated system with other compatible medical devices and electronic interfaces, which may include automated insulin dosing systems, insulin pumps, blood glucose meters or other electronic devices used for diabetes management. Thus, a pump that can communicate with the G6 might have a streamlined FDA review. What’s more, because G6 is now an iCGM, the FDA has also changed its risk category from high risk/Class III to lower risk/Class II, which will likely enable faster FDA reviews of future DexCom CGM innovations and products that are compatible with these devices. The G6 has also been filed with Medicare, with a decision expected later this year.
“The FDA’s recent designation of DexCom G6 as the first iCGM represents a significant catalyst for CGM to be integrated with diabetes management systems like TypeZero’s, as well as smart insulin delivery devices like smart pumps and pens,” a DexCom representative told the Community Blog.
In the next installment of “In MedTech History,” we will highlight another recent advancement in the management of diabetes, Tandem Diabetes Care Inc.’s US launch of its t:slim X2 Insulin Pump with Basal-IQ Technology. The first-of-its-kind Basal-IQ feature helps reduce the frequency and duration of low-glucose events by predicting glucose levels 30 minutes ahead and suspending insulin if they are expected to drop below 80 mg/dL.
#diabetes #CGM #iCGM #glucose #insulinpump #artificialpancreas #AP #T1D #ArnoldKadish #biostator #MilesLaboratory #DexCom #G6 #inControl #TypeZeroTechnologies #MiniMed #Medtronic #AbbottLaboratories #FreeStyleLibre #ThisWeekinMedtechHistory #medicaldevice #medtech #CommunityBlog #MedTech Strategist #tracyschaaf
![]() Trial MyStrategist.com and unlock 7-days of exclusive subscriber-only access to the medical device industry's most trusted strategic publications: MedTech Strategist & Market Pathways. For more information on our demographics and current readership click here.
Trial MyStrategist.com and unlock 7-days of exclusive subscriber-only access to the medical device industry's most trusted strategic publications: MedTech Strategist & Market Pathways. For more information on our demographics and current readership click here.