ARTICLE SUMMARY:
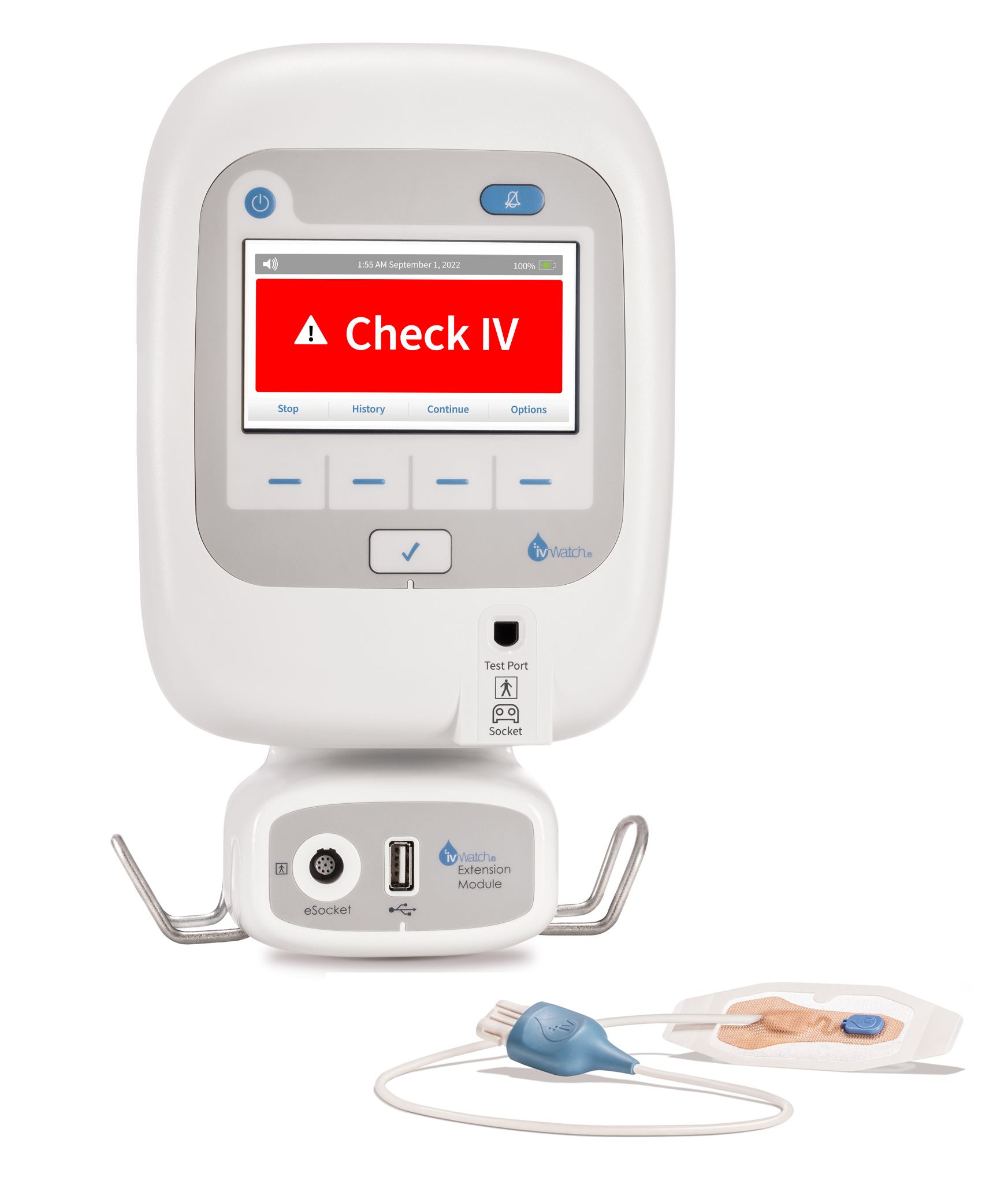
Using optical techniques, ivWatch’s SmartTouch device can detect leaks of a growing range of commonly delivered IV fluids, now including iron, gaining international buzz, not to mention demand, for the technology.
The journey ivWatch has taken from inception to its present state has been incremental, starting with an engineering project in the early 2000s to develop a sensor that could detect IV infiltrations and extravasations. Over time, the team secured SBIR grants totaling $2.8 million to support its efforts and formed the company officially in 2010. That year, Gary Warren joined ivWatch as CEO and brought in a fresh team of engineers, leading to the company’s first FDA clearance for the Model 400 bedside monitor. Fast-forward to the present day, ivWatch’s newest product, the SmartTouch sensor, is gaining traction in a range of use cases, most recently receiving 510(k) clearance in late March 2024 for detecting extravasations of iron sucrose.