ARTICLE SUMMARY:
As the clinical world redefines chronic heart failure populations, new targets for device intervention are emerging and at least four companies are developing novel transcatheter treatments for a type of heart failure that has been increasing in prevalence: heart failure with preserved ejection fraction. Now the most common type of failure, HFpEF offers an emerging device opportunity at the intersection of two high growth medical device markets: heart failure and structural heart.
There are two kinds of chronic heart failure and they present with similar symptoms; breathlessness, fatigue, bloating, and weight gain due to fluid build-up. Both have similar rehospitalization rates—about 30% within 60-90 days of discharge.
But the two kinds of heart failure differ in one important aspect: there are no therapies for one of them, which, astonishingly, accounts for more than half of all chronic heart failure patients. We’re talking about heart failure with preserved ejection fraction or HFpEF (pronounced “HeffPeff”) the newest target for innovation in transcatheter structural heart disease.
For the purposes of treatment, chronic heart failure is broken down into two classes of patients—those in whom the heart fails to pump adequately and those in whom the ventricle fails to fill properly. Heart failure with reduced ejection fraction or HFrEF (formerly known as systolic heart failure), for which there are numerous drug therapies, is a failure of pumping. The left ventricle fails to contract with enough force to supply the body with oxygen and nutrients. In patients with HFpEF (which used to be called diastolic heart failure) the left ventricle ejects blood forcefully but it’s stiff and non-compliant, so it doesn’t fill properly.
HFpEF is defined as the manifestation of classic heart failure symptoms in patients with an ejection fraction greater than 50%, along with other signs particular to the disease such as slow, delayed, and impaired filling of the left ventricle, and increased left atrial pressure during early filling. (But these designations don’t cover all situations—some patients with systolic heart failure also experience diastolic dysfunction and systolic function isn’t always normal in HFpEF patients either.)
At any rate, recently, in clinical and medical device circles, attention has turned to HFpEF, for a couple of reasons. For one, it’s a large and growing problem. Estimates are that 54% of patients with chronic heart failure have this variety and that proportion is likely to grow, because its pathophysiology appears to have more to do with an accumulation of the damages that come with aging (because of underlying conditions like hypertension and coronary artery disease, for example) than a specific cause like a heart attack or valve dysfunction. The second reason is that, as noted, no heart failure therapies have proven to be effective for HFpEF, perhaps because these older patients with co-morbidities are more complicated and heterogeneous than heart failure patients of the other variety.
This complete lack of therapies for a large and very sick patient population has drawn new companies into the market, as has a body of literature based on the gleanings from implantable hemodynamic monitors that became available in the last decade, which found elevated left atrial pressure to be a precursor to episodes of decompensated heart failure and emergency visits to the hospital.
Three start-ups are developing device therapies that shunt blood from the left to right atrium to lower left atrial pressure: the well-funded Corvia Medical Inc. and V-Wave Ltd., discussed below, and the newest entrant, Alleviant Medical Inc. which operates out of the TMCx and JLABS incubators at the Texas Medical Center in Houston (and isn’t ready to speak publicly about its technology). A fourth company, CorAssist Cardiovascular Ltd., is focusing on the impaired filling aspect of HFpEF with a minimally invasive device for improving the compliance of the left ventricle.
These four companies are developing transcatheter products for the heart, and as such are creating an emerging device opportunity at the intersection of two high-growth medical device markets: heart failure and structural heart. The potential market is huge—chronic heart failure affects more than 10 million people in the US and EU; and it’s an economically important market. Later stage heart failure patients are responsible for hospitalization costs of more than $30 billion a year. Strategic investment in the space by cardiovascular companies like Johnson & Johnson, Edwards Lifesciences Corp., and Abiomed Inc. (as well as others who must remain unnamed, according to these start-ups) evidence a growing interest in an adjacent, enormous, and clinically important market opportunity.
Left Atrial Pressure Moves from a Diagnostic Tool to a Treatment Target
Elevated left atrial pressure (LAP) is a harbinger of worsening symptoms and episodes of acute decompensation in heart failure patients, a fact that has been well validated by the advent of implantable hemodynamic monitors from Medtronic plc (the Chronicle product) and Abbott Laboratories Inc. (which acquired CardioMEMS and Savacor, now known as HeartPod). In addition, elevated LAP is associated with increased mortality.
Once it became possible for heart failure clinicians to use the information from such sensors to titrate therapy on a timely basis, these implantable monitoring devices became therapeutic tools, as validated by Abbott’s CHAMPION (CardioMEMS Heart Sensor Allows Monitoring of Pressure to Improve Outcomes in NYHA Class III Heart Failure Patients) trial, which found that the use of implantable hemodynamic monitors, combined with frequent adjustments of the patient’s heart failure medications, reduced heart failure hospitalizations. But, of course, that all depended upon active management on the part of patients, nurses, and clinicians.
In a parallel development, devices to lower LAP emerged and it became obvious to move from using elevated left atrial pressure as a diagnostic tool, to a therapeutic target that would automatically deliver the desired outcome.
Corvia Medical (Tewksbury, MA) is the leading company in the transcatheter treatment of HFpEF and HFmrEF [Heart Failure with Mid Range Ejection Fraction], in terms of having generated compelling clinical evidence by treating the most patients thus far. For Corvia, an understanding of the role of left atrial pressure was combined with decades-old observations that patients with both mitral stenosis, (which causes elevated LAP, and congenital atrial septal defects (ASDs) suffered from fewer symptoms than patients with mitral stenosis without the ASD. ASDs leave a hole (that should normally close shortly after birth) in the septal wall, allowing blood to shunt from the left to right atria.
David Celermajer, MD, PhD, an interventional cardiologist at the University of Sydney, Australia with HFpEF patients under his care, observed that on occasion, when a large ASD was planned to be closed to minimize the risk of right heart failure, left atrial pressure would rise markedly as the sizing balloon was inflated. When the balloon was deflated and the ASD reopened, the pressure would drop. This finding led Celermajer to conceptualize an interatrial shunt that would deliberately allow a small amount of blood to flow from the left to right atrium, to lower LAP when needed (at rest and during activity). Corvia was founded in 2009 to take this therapy from concept to commercialization.
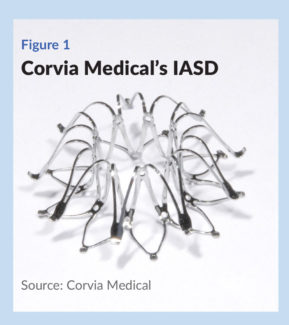 Corvia’s InterAtrial Shunt Device (IASD) creates an 8 mm opening between the heart’s two atrial chambers, a size chosen to achieve the proper flow dynamics without increasing the risk of right heart failure or low forward cardiac output. Company President and CEO George Fazio notes that clinicians only close ASDs of a known clinically relevant size, and do not close smaller ASDs, pointing out that the opening created by the IASD is smaller than septal defects that warrant closure. The IASD resembles a low-profile stent; when deployed it is flat against tissue on the left side and convex on the right, to accommodate septal walls of varying thickness ( see Figure 1).
Corvia’s InterAtrial Shunt Device (IASD) creates an 8 mm opening between the heart’s two atrial chambers, a size chosen to achieve the proper flow dynamics without increasing the risk of right heart failure or low forward cardiac output. Company President and CEO George Fazio notes that clinicians only close ASDs of a known clinically relevant size, and do not close smaller ASDs, pointing out that the opening created by the IASD is smaller than septal defects that warrant closure. The IASD resembles a low-profile stent; when deployed it is flat against tissue on the left side and convex on the right, to accommodate septal walls of varying thickness ( see Figure 1).
The device is navigated from the femoral vein, through the inferior vena cava—routine transcatheter techniques for interventional structural heart clinicians placing ASD or PFO closure devices or left atrial appendage occlusion devices—and directed to the atrial septum, into which a needle puncture has been made. The device is advanced over a guidewire until the tip is positioned in the left atrium, and is sequentially deployed, fist in the left and then in the right atrial side. The delivery system and guidewire are removed, and the procedure is complete.
To date, Corvia has raised $58 million and gained CE mark for its IASD based on a 64-patient open-label, single arm study called REDUCE LAP-HF. It has completed REDUCE LAP-HF I, a 44-patient randomized, 1:1 sham-controlled, double-blinded clinical trial (conducted at 22 centers in the US, Europe, and Australia) and is well into its 608-patient randomized, 1:1 sham-controlled, double-blinded pivotal trial, REDUCE LAP-HF II.
REDUCE LAP-HF I, which was rigorously conducted, produced definitive mechanistic results and additional encouraging clinical results. Investigators randomized patients 1:1 to sham control or treatment with IASD. In the control group, patients underwent a procedure involving femoral venous access followed by intracardiac or transesophageal echocardiography, but no IASD.
One month results demonstrated that pulmonary capillary wedge pressure (a surrogate measure of left atrial pressure) was significantly lower in the treated group. At one year, heart failure hospitalization rates were lower in the treated group (.22 events per patient year for IASD compared to .63 for the control). On the endpoint of reduction in NYHA classification, treated patients experienced a one class improvement at one year while patients receiving the control procedure experienced no improvement.
Although there is a long way to go, much has changed since Corvia first entered the field. “When we would go to heart failure meetings in 2010 and talk about creating an interatrial shunt, people would say ‘You are creating a hole? We need to close that hole,’” says Fazio. “While there was human experience with having a natural shunt between the left and right atrium, there was no human experience with a created shunt. But our theory has held up, and there is consistency between our 11-patient pilot study, our 64-patient CE mark study and our 44-patient randomized study with respect to safety and durable clinical benefit.” Fazio adds, “We’ve been at this since 2009, and things are different now,” he says. “There are now packed, standing-room-only sessions dedicated to HFpEF at all the heart failure and large interventional cardiology meetings.”
The field is still looking for large rigorous clinical trials, and Corvia is in the process of meeting that demand with its 608-patient pivotal trial, now taking place at close to 100 sites in the US, Europe, and Australia. Sanjiv Shah, MD from Northwestern University and Martin Leon, MD from New York Presbyterian/Columbia University are the principal investigators. [ Editor’s Note: As this article was going to press, Corvia announced an agreement with Edwards Lifesciences. Edwards will provide $35 million in funding for the completion of the REDUCE LAP-HF-II clinical trial, and gets an exclusive option to acquire Corvia at a later date.]
V-Wave: Treating a Pathway Common to All Heart Failure
Like Corvia, V-Wave Ltd. (Caesarea, Israel) is focusing on reducing left atrial pressure with an implantable interatrial shunt, but its approach and its market strategy are a bit different. The company is currently conducting a pivotal study enrolling 500 patients with symptomatic heart failure with both HFpeF and HFrEF in the NYHA Class III and the ambulatory Class IV populations. Says Neal Eigler, MD, CEO, an interventional cardiologist and a serial medical device entrepreneur (he is a founder of Progressive Angioplasty Systems, Conor Medsystems, and Savacor), “we made the decision from the get-go to approach both simultaneously, since we knew from hemodynamic monitoring studies that lowering left atrial pressure in both groups of patients is an effective intervention for keeping them out of the hospital, and also, possibly, for improving life expectancy.”
V-Wave was founded in 2009 around a concept published by Gad Keren, MD, who has been professor of cardiology at Tel Aviv University’s Sackler School of Medicine since 1994. In 2015, the company was looking for a pathway to regulatory approval in the US and commercialization, and brought on Eigler as CEO. Eigler was a logical choice; he was intimately familiar with the problem and the population as the founder of Savacor, which developed an implantable hemodynamic monitor and an interface for patients to use to titrate their own heart failure therapies on a daily basis, based on algorithms built into the system. St. Jude Medical (now Abbott) bought the start-up in 2005 for $50 million.
After Eigler’s arrival, V-Wave raised a $28 million Series B round that brought in two strategic investors—Johnson & Johnson Development Corp. (JJDC), which led the round, and Edwards Lifesciences—as well as TriVentures Inc., Pura Vida Investments, BioStar Ventures, BRM Group and Pontifax. A Series C round led by Deerfield Management closed in April 2018, bringing in $70 million from existing investors and new investors Endeavour Vision, Quark Venture, Aperture Venture Partners, and Israel Secondary fund.
 The V-Wave Shunt is an hourglass-shaped implant on a nickel titanium alloy frame. The frame is encapsulated on its interior and exterior surfaces with ePTFE, an approved antithrombogenic polymer. The diameter of the device is about 5 mm at its “waist” ( see Figure 2) . Delivery of the device makes use of the femoral vein access and transseptal cardiac catheterization that is familiar to interventionalists delivering transcatheter structural heart or electrophysiological treatments.
The V-Wave Shunt is an hourglass-shaped implant on a nickel titanium alloy frame. The frame is encapsulated on its interior and exterior surfaces with ePTFE, an approved antithrombogenic polymer. The diameter of the device is about 5 mm at its “waist” ( see Figure 2) . Delivery of the device makes use of the femoral vein access and transseptal cardiac catheterization that is familiar to interventionalists delivering transcatheter structural heart or electrophysiological treatments.
“Interatrial shunting could supplement current therapies by directly regulating cardiac pressures and volumes. The V-Wave approach has been physiologically validated and is self-regulating,” Eigler says. As left heart pressure rises, the amount of shunting increases and this shift of volume from the left to right atrium then lowers left heart pressures. As noted, controlling left heart pressures has been shown to improve patient symptoms and clinical outcomes.
According to Eigler, the architecture of V-Wave Shunt offers several advantages. First, interventionalists are able to use the patient’s anatomy to register the location of the device during deployment. “It makes deployment more certain and relatively simple. We have implanted these in almost 60 patients now, and they have all gone into the right location on the first try.” The shape—wider entry and exit funnels on both sides of the atria, and a narrow waist in the septum—allows the hole in the septum to be only as large as it needs to be to provide the right fluid dynamics—as to the rate and volume of blood flow. Eigler also points out that the septal wall is important real estate that needs to remain passable for transcatheter mitral valve repair or other interventions, so a shunt device should not be too large.
First human results with an earlier iteration of the shunt were published in 2018. The first 38 consecutive patients were successfully implanted during a minimally-invasive procedure that generally took about an hour and patients were discharged to their homes the next morning. There were no device-related adverse events. At one-year follow-up, mortality and hospitalization due to worsening heart failure were low and there were significant improvements in symptoms, exercise capacity, and quality of life measures.
In October 2018, V-Wave commenced enrollment for RELIEVE-HF, a patient and observer blinded randomized, controlled pivotal study of 400-600 NYHA Class III and ambulatory Class IV patients at up to 50 centers in North America and 25 centers across Europe and Israel. The recent $70 million Series C round will support the trial to completion.
“We are doing the best science we can,” says Eigler, to validate the therapy for the clinicians that will eventually be choosing it. While structural heart disease cardiologists and electrophysiologists are known to be early adopters of new technology, “heart failure specialist clinicians tend to be more conservative and want to see more quality data, especially from randomized trials, before they’ll change their practice or refer patients for new technology. Good science is also the best way to get third-party payors to pay for it.”
There is a marked need to have better therapies for these Class III and Class IV heart failure patients, says Eigler, who estimates that V-Wave’s market in the US is 1.2-1.4 million patients (out of a total of 6.2 million chronic heart failure patients in the US). “Not only are there a million-plus patients in the US and five to six million worldwide that could feel better, exercise more, stay out of hospitals, and have a better quality of life with a treatment like this, there are societal costs,” specifically, some $30-$40 billion in direct costs related to hospitalizations, just in the US.
According to one study, the mean per-patient cost for a heart failure hospitalization is $14,631 and the 30-day readmission rate is 22.3%, rising to 40.2% at 90 days (Kilgore, M. et al, “Economic burden of hospitalizations of Medicare beneficiaries with heart failure,” Risk Management Healthcare Policy, May 10, 2017).
“From both the societal and healthcare economics standpoints, being able to keep these patients out the hospital is extremely important and worth this kind of investment.” And, without an effective therapy, the future looks bleak for patients with advanced heart failure. “When you go out three or five years, the prognosis for these patients is worse than for most cancers. You have a group of underserved patients and you also have the opportunity to have a favorable benefit versus cost.” The winds are blowing in the right direction, Eigler says. “All we need now is the scientific proof that the device can do what we think it has the potential to do.”
CorAssist Cardiovascular: A Transcatheter Treatment for the Left Ventricle
As noted, elevated left atrial pressure is a pathway to heart failure that’s common to both HFpEF and HFrEF, and that’s why the majority of the new companies have focused their therapeutic strategies on lowering pressure in the left atrium. But CorAssist Cardiovascular Ltd. (Haifa, Israel) is developing a transcatheter therapy to directly address what it sees as the hallmark of HFpEF; the inability of a stiff and non-compliant left ventricle to relax and fill properly. CorAssist’s CORolla implant is delivered by a transapical approach into the left ventricle, where it augments the function of the left ventricle during diastole.
The company was founded in Israel in 2002 by three clinicians. Yair Feld, MD, PhD, is an interventional cardiologist at the Rambam Health Care Campus in Haifa. He’s also the founder of four other medtech companies, two of them in heart failure (Paragate Medical and Nephera Ltd.). Co-founder Yotam Reisner, MD, PhD, has managed R&D and/or clinical research for a number of medical device companies, including Itamar Medical, Enopace Biomedical, Nephera, and EPD Solutions. The third founding member, Shay Dubi, who has an MD and PhD in cardiovascular physiology, was previously a senior executive at four medical device companies, including MValve Technologies Ltd., which operates in the transcatheter mitral valve market. He is also a founder of two medical companies, JetPrep and Best Med Opinion Ltd. Amir Elami, MD, who joined early on, is a cardiac surgeon at Hadassah Medical Center in Jerusalem.
Feld says he was a medical student in 2000 when he was surprised, during the course of a lecture, to learn about the features of HFpEF and the great extent of the disease. “The lecturer said that the problem in many patients was that the heart could still contract, but it couldn’t relax, so I thought about putting a coil in to help it relax.” He sought out his medical student friends (the afore-named founders), and together with Dr. Elami, they came up with a concept of a self-expanding spring to offload the energy needed to help the heart open and receive blood in diastole. They built a prototype, and were accepted into an incubator of the Israel Innovation Authority.
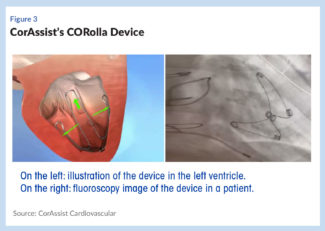 For the first 10 years, the company focused on an epicardial version of the coil, and got it as far as clinical trials, which Feld says did demonstrate proof-of-principle of the approach. However, the surgery to implant it was complicated and lengthy, so the product was shelved in favor of the current device, the CORolla, which, as noted is delivered via a transapical approach, as are many TAVR (transcatheter aortic valve replacement) products ( see Figure 3).
For the first 10 years, the company focused on an epicardial version of the coil, and got it as far as clinical trials, which Feld says did demonstrate proof-of-principle of the approach. However, the surgery to implant it was complicated and lengthy, so the product was shelved in favor of the current device, the CORolla, which, as noted is delivered via a transapical approach, as are many TAVR (transcatheter aortic valve replacement) products ( see Figure 3).
The idea is to transfer energy from the systolic phase of the cardiac cycle (when the ventricle is contracting) to the diastolic phase, says Feld. An elastic metal spring implanted in the apex of the left ventricle absorbs energy during systole. During diastole it applies direct expansion forces to the ventricle wall and septum to improve the dynamics of relaxation and filling.
CorAssist is currently conducting its first-in-man study of 10 patients. The first standalone patient was implanted in 2017, and the company foresees completing the study in the next 18-24 months. Results have been positive so far, Feld says, and while it’s too early to discuss specifics, the company has learned that the CORolla does more than just open up the left ventricle. “The device does two major things,” he says. “One, obviously, it improves the compliance. By pressing on the left ventricle, by helping relaxation, we reduce the pressures that are needed for the heart to fill. So that reduces the pressures in the lungs and lung congestion.” But there is an additional potential benefit. “When you reduce the pressure in the left ventricle during the diastolic phase, you improve the coronary perfusion pressure, and that improves blood supply to the heart.” Feld notes that many patients with HFpEF also have microvascular disease. “If we can improve the blood supply, we can improve the pathophysiology that leads to a reduction in compliance.”
To date CorAssist has raised about $17 million privately from a group of investors that includes Aurum Ventures, MKI, Ofer Hi-Tech Group, Yozma Management, Evergreen Venture Partners, Argonaut Ventures, and Abiomed Inc.
Next steps for the company are completion of the ongoing human pilot trial (which is funded by Abiomed), after which it plans to move onto full-scale clinical trials that support CE mark and PMA approval in the US. Future plans also include the development of transfemoral products.
A Shift in Heart Failure Treatment Paradigms
If HFpEF has become an attractive target for medical device innovation, it's primarily because, as noted, there are no therapies and it affects so many people. But these new approaches might also signal a shift in the way heart failure will be understood and treated in the future.
As discussed earlier, treatments for chronic heart failure have generally revolved around the measurement of ejection fraction, i.e., have categorized patients as having a disorder related to either heart pumping or heart filling. But heart failure is ultimately a function of damage to the myocardium and the geometric remodeling that occurs because of that unhealthy tissue.
In HFpEF, the pathway to that outcome appears to be particularly complex. HFpEF patients are older and it affects more women than men. Because it’s thought to be a function of all the energetic and cellular assaults that come with aging, which are caused by hypertension, diabetes, obesity, obstructive sleep apnea, and a host of other disease and processes that affect the elderly, it’s opened up a whole new area of research that might ultimately result in new treatment strategies for heart failure. Renal denervation, for example, which is primarily being looked at as a treatment for drug-resistant hypertension, also has the potential to help HFpEF, not only by controlling hypertension, but by suppressing excess sympathetic activity. Several studies are looking at the potential for renal denervation to influence ventricular structure and diastolic function.
This category is by no means going to be an overnight sensation; these are devices that are implanted in the heart and represent a long and expensive undertaking. Start-ups will be required to fund very large 500-600 patient, randomized, controlled clinical trials that take years to enroll. The two leaders in the space, Corvia Medical and V-Wave have done so, and it appears that when the studies are finished, they’ll be adequately powered to do the sub-group analyses that could help the clinical worlds and industry understand heart failure better, and how and where devices can intervene to improve outcomes.

