ARTICLE SUMMARY:
The multibillion-dollar molecular diagnostics market is one of the fastest growing sectors in the life sciences. Why should the medical device industry pay close attention?
A new white paper from Enterprise Analysis Corp (EAC), a well-established market research and strategy firm, quantifies in concrete terms the stellar growth of molecular diagnostics and the sub-sector’s important role in the clinical laboratory. The numbers are impressive, as is the underlying story, especially given that the first FDA-approved tests based on PCR analysis came to market in the mid-1990s and that in 1998, the market size was only $500 million.
EAC estimates that the global molecular diagnostics market as of 2017 was $6.6 billion, $7.2 billion in 2018, and, with a CAGR of 8.5%, it will reach an estimated $9.9 billion by 2022—making it one of the fastest growing sectors in life sciences (can you think of another medical device subsector in that league?) Advances in a mix of scientific disciplines—notably nanotech, genomics, and drug development—are behind some of the excitement, as well as external drivers, which include FDA regulatory shifts that have led to the launches of important new therapies, and, more mixed, CMS’s recent reimbursement policies. That doesn’t include the wave of consumer genomic tests that are creating a bottoms-up groundswell of interest in a sub-set of molecular diagnostics that is rapidly becoming part of our culture.
So why should the medical device industry pay close attention?
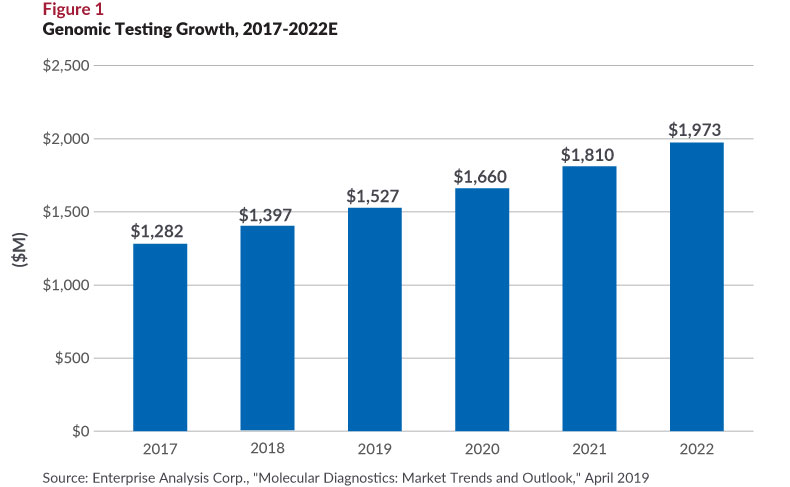
There are dozens of FDA-approved molecular tests for multiple indications on the market—and hundreds more, if not thousands, if the count includes laboratory developed (LDTs) and research-use-only tests (RUOs). Infectious disease (not cancer!) is the largest subsector at $4.1 billion, or 62% of the total market, followed by the genomic segment (cancer, HLA testing, pharmacogenetic tests, and genetic diseases), at about 20% of the total, according to EAC. Of all the subsectors, genomics is growing faster, driven by high-value cancer diagnostics and the conversion of PCR-tests to next-generation sequencing (see Figure 1.)
No better way to highlight the importance of these trends to medtech exists than to point to surgical oncology, which is already heavily impacted by results of genetic tests; just one example, the American Society of Breast Surgeons earlier this year announced new guidelines calling for all patients diagnosed with breast cancer to be given an opportunity for genetic testing, as opposed to the previous recommendations that restricted testing to certain age groups and sub-types of cancer. “Breast surgeons are often the first physicians who interact with patients after they are diagnosed with breast cancer, and they can guide patients through the testing and interpretation process,” Walton Taylor, MD, ASBrS president said in a statement. But in the future, other high-volume specialties will be impacted to varying degrees, as well—cardiology comes to mind, although orthopedics may be further afield. We’ll be writing more about this in upcoming issues of MedTech Strategist.
More broadly, leading healthcare systems are transforming their precision medicine strategies beyond very specific indications in select specialties into systemwide initiatives—a feat that requires individualized patient treatment based on sophisticated data analytics. Among the core components enabling their efforts are advances in molecular testing technologies. These innovations lower costs and automate complex steps in the laboratory work flow process. [As part of this trend, we’ve written about NorthShore University Healthsystem’s program to bring genetic testing to primary care physicians, and most of the major academic medical centers taking similar steps, albeit from different angles.] As the EAC report points out, highly complex testing previously done by specialized staff is consolidating in core laboratories, where routine testing is done at a single site by lesser trained lab technicians. At the complex end, next-generation sequencing is being positioned to play a bigger role in more treatment categories.
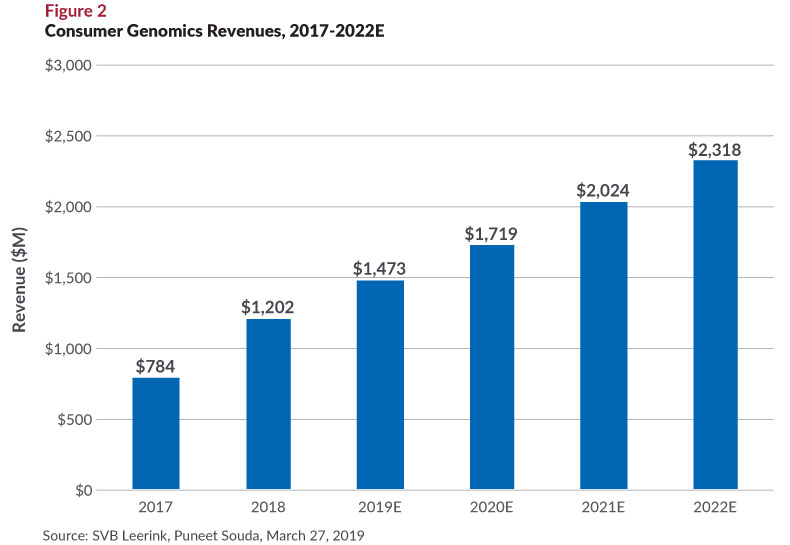
At the same time, more molecular testing is moving to the point of clinical care and even beyond that to the consumer setting (see Figure 2). Rapid, sample-to-answer capabilities are enabling more POC testing in the doctors’ office, largely focused on influenza and Strep-A. This is only a small market now, but poised to grow as more systems qualify for CLIA-waivers—the regulatory status that enables them to be operated without specially trained staff.
And proactive consumers are an increasing part of the equation. Call it democratization of molecular testing, it comes with multiple, high-stakes challenges. Maintaining quality comes to mind. And, as is clear from attending any medical meeting on genetics these days, at the top of the list is an unresolved qualitative hurdle: the education and communications gap. If these issues are pushed under the rug, the qualitative challenges may trump the stellar quants.
“The field is moving so rapidly, physicians, particularly primary care doctors, can’t keep up,” points out Robert Michel CEO and founder of The Dark Intelligence Group, a business intelligence company focused on clinical laboratories and pathology. “Walking into specialists’ and increasingly primary care physicians’ offices are a growing number of patients who order their own genetic tests and are getting ahead of their doctors in attempting to understand what these tests mean for their own health. How quickly hospitals set up resources to address this in the clinical care continuum is happening more slowly and at a limited number of institutions.” The Precision Medicine Institute Symposium 2019, a summit his company is organizing on May 2 and 3 in New Orleans, showcases some of the first movers in systematizing this field, such as Vanderbilt University Medical Center and Geisinger National Precision Health. Even these systems are still trying to dial in what it will take, the size of the demand, and what kind of medical and scientific expertise is needed on staff, Michel says.
#moleculardiagnostics #genomics #precisionmedicine #MedicalDevice #CommunityBlog #medtech #medicaldevices #MedTechStrategist
![]() Trial MyStrategist.com and unlock 7-days of exclusive subscriber-only access to the medical device industry's most trusted strategic publications: MedTech Strategist & Market Pathways. For more information on our demographics and current readership click here.
Trial MyStrategist.com and unlock 7-days of exclusive subscriber-only access to the medical device industry's most trusted strategic publications: MedTech Strategist & Market Pathways. For more information on our demographics and current readership click here.