ARTICLE SUMMARY:
Diseases of the deep veins can be deadly, but this clinical segment has been an innovation backwater. Things are changing: the first venous stents were approved in 2019, and several start-ups are addressing unmet clinical needs with devices purposely built for veins, not borrowed from the arterial side of the anatomy.
Not so very long ago, a medical device entrepreneur attended a clinical conference on venous disease to learn about the field, and was surprised by the decidedly low-tech nature of current therapies. “It looked more like JC Penney’s than a clinical conference,” he said, “lots of mannequin legs wearing socks.”
He was of course referring to compression stockings, the mainstay conservative therapy offered to many chronic venous insufficiency patients.
It’s true that venous disease has lagged other specialties in the development of interventional tools that address underlying causes and not just symptoms, despite the fact that the clinicians that treat the veins are the same ones that treat peripheral artery disease with endovascular revascularization tools—vascular surgeons, interventional radiologists, and interventional cardiologists. Clinicians have had little to offer these patients, which far outnumber those with peripheral artery disease. Some 30 million people in the US suffer from venous conditions.
But if venous disease hasn’t seen much innovation, it could be due to the assumption that it’s less immediately threatening than coronary artery disease, and not likely to lead to amputation as rapidly as peripheral artery disease.
That level of benignity might be true for much of superficial vein disease, which manifests as spider, reticular, and varicose veins, visible at the surface of the body. Superficial venous reflux has been regarded as somewhat of a cosmetic or quality-of-life indication that has supported the development of a billion-dollar-plus semi-consumer/semi-medical market served by endovenous technologies for ablating diseased veins.
But both superficial and deep venous reflux can have more serious consequences, causing venous leg ulcers that result in open sores that don’t heal for months or even years in some cases. In the US, 500,000-600,000 people suffer from venous leg ulcers, the majority of them due to underlying chronic venous insufficiency, and the annual cost of treating them is estimated at $2.5-$3.5 billion.
In fact, venous disease can be quite serious; deadly obstructions of the venous system can occur; deep vein thrombosis (DVT) can throw clots into the pulmonary artery, and occlusions in the deep veins of the pelvis can cause painful swelling in the extremities, patient immobility, and blood clots.
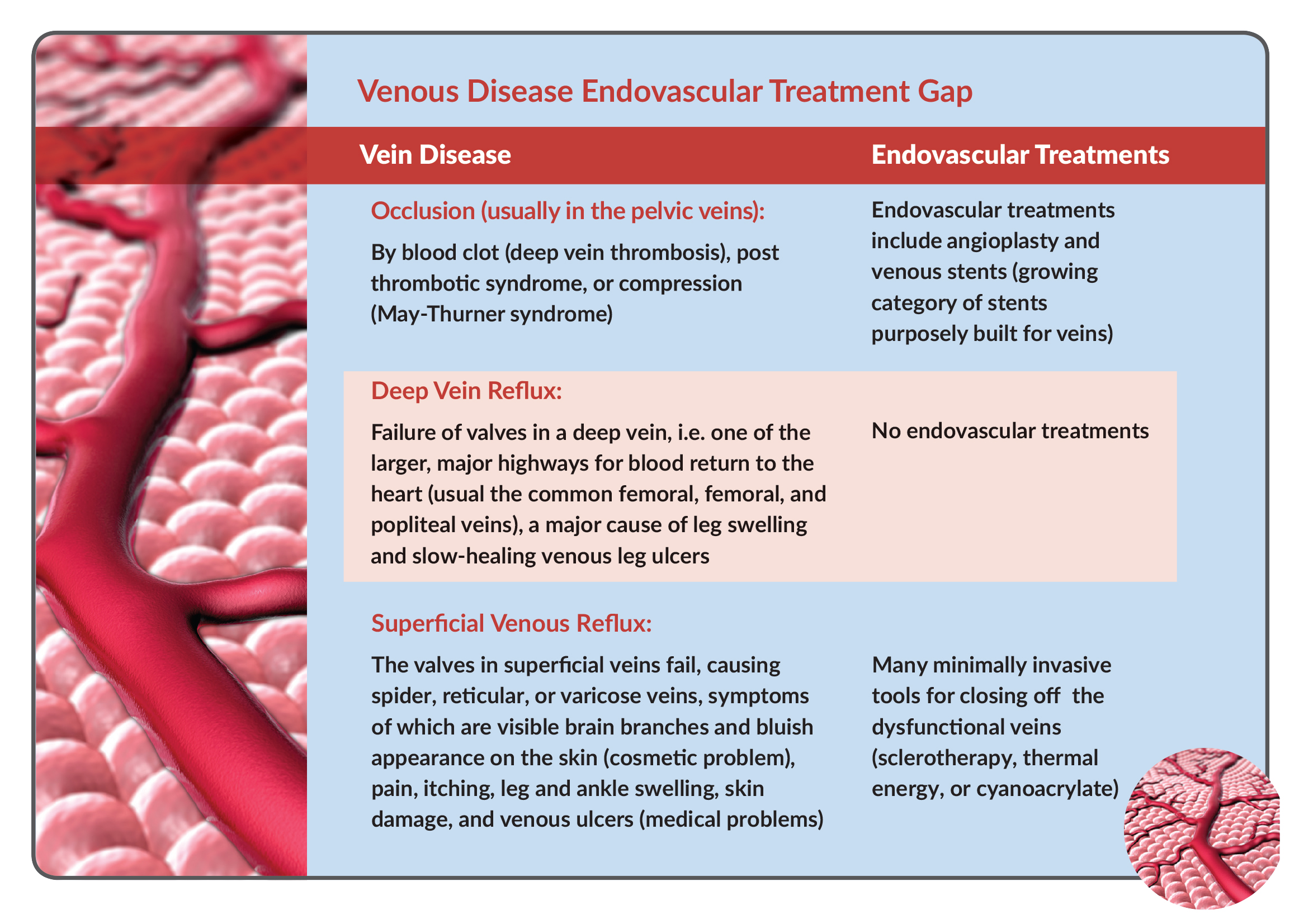 These more serious conditions would seem to be amenable to endovascular revascularization technologies from a medtech industry adept at serving coronary and peripheral arterial disease, and indeed, clinicians have repurposed endovascular tools (on an off-label basis) to applications in the venous system as the need has arisen.
These more serious conditions would seem to be amenable to endovascular revascularization technologies from a medtech industry adept at serving coronary and peripheral arterial disease, and indeed, clinicians have repurposed endovascular tools (on an off-label basis) to applications in the venous system as the need has arisen.
But there are signs that “interventional vein” is up-and-coming, as companies increasingly develop devices specifically built for the unique requirements of the venous system.
An Endovascular Specialty’s Time Has Come
This year has seen the approval of two venous stents; first, in March 2019, was the Venovo, from Becton Dickinson (BD), for the treatment of the Iliofemoral vein segment, based on results of its VERNACULAR clinical trial. Two months later, positive results from the VIRTU study supported FDA approval for the VICI venous stent from Boston Scientific Corp., which the strategic gained last year when it acquired the remaining 75% of Veniti that it didn’t already own, for $108 million in cash up front and $52 million contingent upon US approval of the VICI.
These are not just up-sized arterial stents repurposed for vein anatomy, they’re the first FDA-approved devices designed specifically for veins. Meanwhile, Cook Medical Inc. has been selling its Zilver Vena Venous Self-Expanding Stent in Europe since 2010 and Medtronic plc is not far behind; its Abre Venous Self-Expanding Stent system received CE mark approval in April 2017.
Medtronic got a head start on its venous device portfolio when, in 2014, Covidien acquired Sapheon, the developer of VenaSeal, a novel approach to endovascular vein ablation. And Boston Scientific gained a portfolio of venous devices from its acquisition of BTG plc, which closed in August 2019. Vascular specialist Penumbra Inc. has also expanded into veins, with a mechanical thrombectomy device for the treatment of DVT.
In addition to the major strategics looking to fill pipelines of vein-dedicated products, we now have a number of well-funded, venture-backed medical device start-ups focused on serving unmet clinical needs in chronic venous insufficiency an other vein problems (see Figure 1). We interview three in this article: Vesper Medical Inc., the developer of an innovative venous stent; InterVene Medical Inc., which is recreating vein valves to address the root cause of most vein diseases, and Venari Medical, which has a more benign office-based treatment for superficial vein reflux. Has the time finally come for venous devices?
 “We think so,” says Bruce Shook, CEO of Vesper Medical and Intact Vascular Inc. “Historically, the arterial market has dominated everybody’s interest. The arterial business is relatively mature now, and as companies look around for new growth markets, deep venous disease has popped up on their radar. Just to give you some numbers, the prevalence of patients with deep venous disease in the US exceeds five million and 500,000 people are being added to the pool each year. It’s a significant disease state.” And, here, it seems, tools borrowed from the arterial side just aren’t going to cut it.
“We think so,” says Bruce Shook, CEO of Vesper Medical and Intact Vascular Inc. “Historically, the arterial market has dominated everybody’s interest. The arterial business is relatively mature now, and as companies look around for new growth markets, deep venous disease has popped up on their radar. Just to give you some numbers, the prevalence of patients with deep venous disease in the US exceeds five million and 500,000 people are being added to the pool each year. It’s a significant disease state.” And, here, it seems, tools borrowed from the arterial side just aren’t going to cut it.
Veins are Very Different from Arteries
Intact Vascular(Wayne, PA), developer of the Tack Endovascular System for peripheral artery disease, serves vascular surgeons, interventional cardiologists, and interventional radiologists. (MedTech Strategist will focus on Intact Vascular in an upcoming issue.) In conversations with its customer base, says CEO Bruce Shook, it became apparent that there is increased interest in treating deep venous disease. “At the conferences we attended, more and more time was devoted to the treatment of deep venous disease, which is a fairly new phenomenon for this group of physicians.” As noted, historically, these diseases were treated medically with anticoagulants and compression stockings, which dealt with symptoms, but did not address the underlying problem. “The idea that these operators could intervene on deep venous disease and really fix the problem was gaining some traction,” Shook says.
Scanning the horizon for endovascular tools that could treat the deep veins, Intact’s management found tools repurposed from arterial applications delivering some efficacy but subject to drawbacks in terms of deliverability and durability, essentially because arteries are very different from veins in function and physiology.
Arteries, which carry oxygenated blood out of the heart to tissues in the rest of the body, have thick, muscular walls and can contract and constrict according to blood volume. “Think garden hose,” says Shook. Veins, which carry blood from tissues of the body back to the heart, are larger (vein diameters can be up to 20mm whereas arteries in the leg rarely have a diameter greater than 5mm) and are thin-walled. “Think flexible drinking straw,” says Shook. Veins have valves to keep blood flowing towards the heart, which arteries don’t need because pressure from the heart keeps blood flowing in one direction.
Venous disease also differs from arterial disease in many respects, Shook points out. Arterial disease is due to the accumulation of insults—smoking, diabetes, obesity, sedentary lifestyle, high fat diet, etc.—and affects older patients. Venous disease, which is due to family history, extended periods of sitting or standing, obesity, and pregnancy, skews to younger patients and women. And blood clots aren’t the only manifestation of vein disease; others are reflux and the pooling of blood, venous skin ulcers and, in severe cases, the formation of fibrous webbing inside the veins that can significantly obstruct blood flow.
It thus makes sense that devices designed for arteries don’t necessarily translate well to veins. “We saw an opportunity to develop a family of deep venous stents purposely designed for the treatment of iliofemoral vein segments,” says Shook.
Vesper Medical: A Purpose-Built Stent for the Venous System
Intact Vascular’s management and investors decided to repurpose Intact’s skills in developing endovascular tools for peripheral artery disease for a new company, Vesper Medical, which would develop venous devices. Founded in 2016, Vesper Medical has its own mission and independent financing, but shares management and other services with Intact Vascular on a contract basis. (For more on the Intact/Vesper spin-out structure see the upcoming issue of MedTech Strategist.)
Vesper’s Series A round in 2018 raised $10.5 million from NEA (New Enterprise Associates) and Quaker Partners. The $37 million Series B round, announced in May 2019, was led by Vensana Capital and Gilde Healthcare, with the participation of NEA and Quaker.
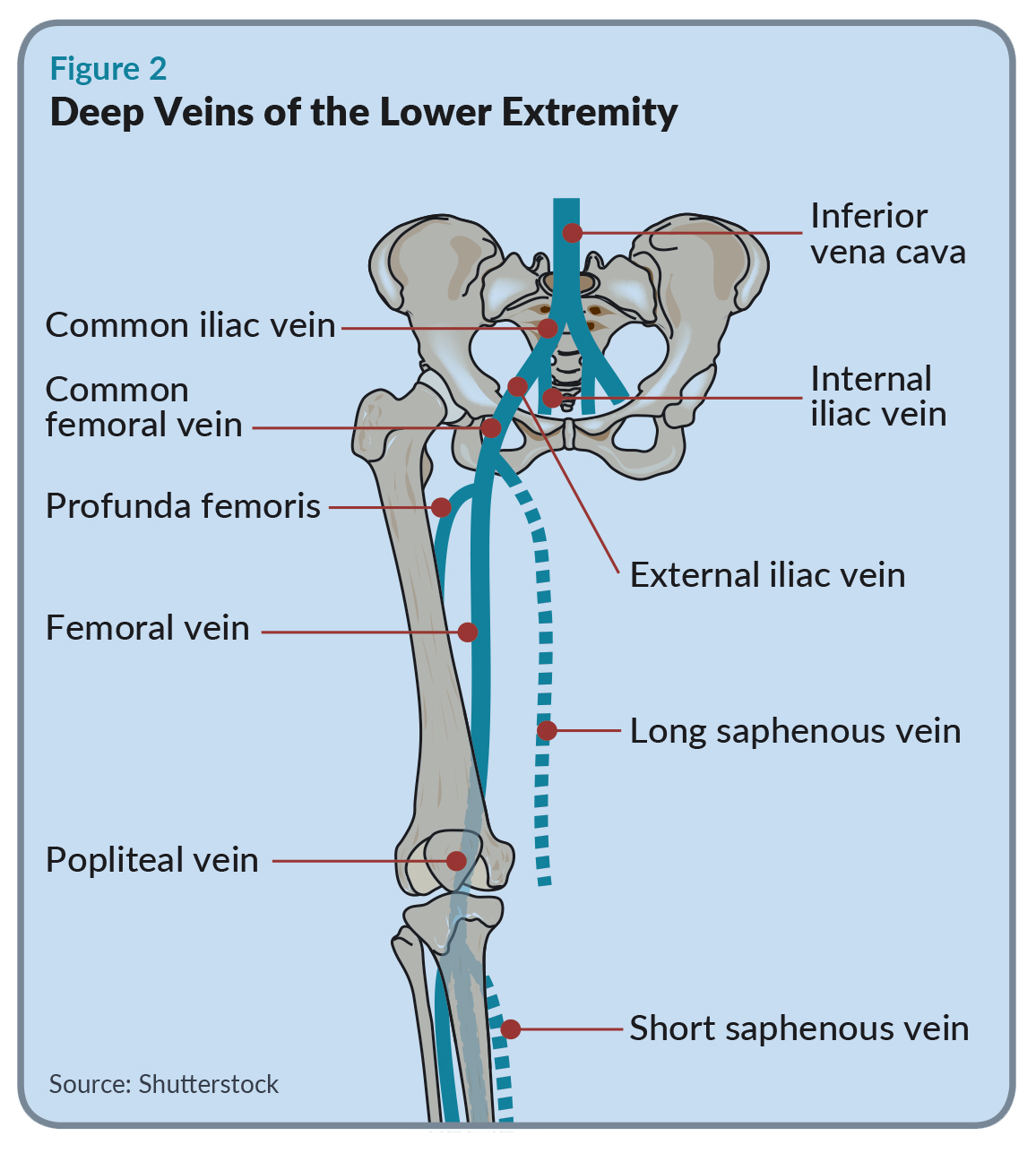 Vesper Medical has developed a family of venous stents specifically designed for treating the iliofemoral vein segments. The iliac veins in the pelvis are large vessels that extend from the inferior vena cava (IVC) down to the inguinal ligament (the crease in your groin) where they transition to the slightly smaller femoral veins (see Figure 2).
Vesper Medical has developed a family of venous stents specifically designed for treating the iliofemoral vein segments. The iliac veins in the pelvis are large vessels that extend from the inferior vena cava (IVC) down to the inguinal ligament (the crease in your groin) where they transition to the slightly smaller femoral veins (see Figure 2).
When the iliofemoral vein segment becomes occluded, venous drainage stops and the affected leg can swell up to two to three times its normal size (see Figure 3). This is painful as well as dangerous, since it sets the stage for the development of clots elsewhere as well as venous ulcers on the leg, which take weeks or even years to heal.
The underlying conditions that set up such occlusions include deep vein thrombosis, which is a clot that forms in a vein; post-thrombotic syndrome, the formation of clots or fibrous webbing in the aftermath of a DVT, or May-Thurner syndrome, whereby the right common iliac artery lies in a location that constricts the left common iliac vein (a condition that can affect young to middle aged women).
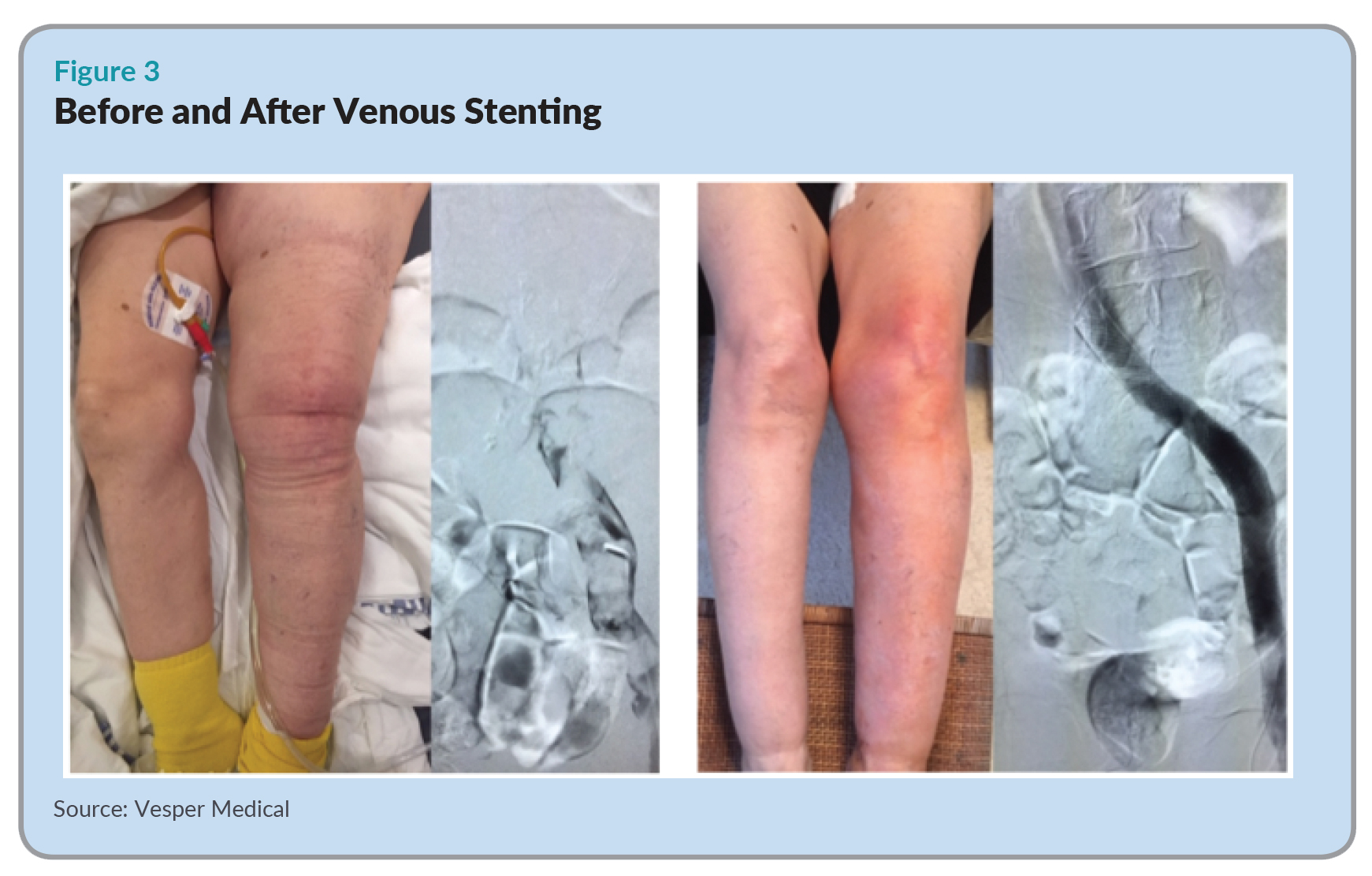 Vesper Medical’s mission was to design a venous stent system that would meet the needs of that particular vein segment, which has different requirements along its length from the top at the IVC to the distal segments further down the leg. According to Shook, “You want a stent that can withstand crush at the top part of the iliac vein [to withstand the pressure of the over-riding artery in May-Thurner syndrome, for example], but as you move down the vein, flexibility and durability become more important.” A lot of flexion occurs near the inguinal segment, when people climb stairs, for example. “So the stent needs to be able to flex, without compressing, and to be super durable,” says Shook.
Vesper Medical’s mission was to design a venous stent system that would meet the needs of that particular vein segment, which has different requirements along its length from the top at the IVC to the distal segments further down the leg. According to Shook, “You want a stent that can withstand crush at the top part of the iliac vein [to withstand the pressure of the over-riding artery in May-Thurner syndrome, for example], but as you move down the vein, flexibility and durability become more important.” A lot of flexion occurs near the inguinal segment, when people climb stairs, for example. “So the stent needs to be able to flex, without compressing, and to be super durable,” says Shook.
The Vesper Duo Venous Stent system consists of a hybrid stent (hybrid, because its mechanical properties change from top to bottom) and a separate extension stent. The Vesper Duo is highly crush resistant at the top and the stent geometry transitions to permit high flexibility as it passes through the pelvis. The extension stent, which is flexible and durable, can be added on to cover the leg further down if necessary. “Every patient is different, so we make both the hybrid stent and the extension stent in various diameters and lengths. It gives the operator the ability to tailor the stent to the individual,” says Shook.
Vesper Medical anticipates beginning its pivotal clinical trial in the first half of 2020, and Shook expects that enrollment (of about 170 patients) will take approximately one year. Investigators will then complete the 12-month follow-up required by the FDA, after which the company hopes to find itself in an emerging market with many positive attributes. Reimbursement already exists for venous stenting, for example, and vascular surgeons, interventional radiologists and interventional cardiologists are already looking for such products. Finally, it’s early but not too early. As noted, BD and Boston Scientific recently introduce the first venous stents to the US market, and others are coming from Cook Medical and Medtronic.
Superficial Vein Disease is a Big Industry
When most people think of vein disease, they think of spider veins and varicose veins, visual signs of underlying chronic venous insufficiency, which is due to the breakdown of vein valves. Spider veins are more of a cosmetic problem; varicose veins, when severe enough, are treated, and often reimbursed, like a medical condition, because they can be painful and can result in immobility and venous ulcers.
Chronic venous insufficiency affects 20-40% of the adult population in the US and a large service industry in the form of chains of vein centers has arisen to treat patients with superficial vein disease on an outpatient basis.
The therapeutic goal for these patients is to close off the diseased vein, preventing blood pooling in the legs and allowing it to circulate in healthy veins. Four modalities are available today to accomplish this: surgical stripping (the removal of the damaged vein by an open surgical procedure); endovenous thermal ablation, which uses laser or radiofrequency (RF) energy to destroy the vein; sclerotherapy, the injection of a toxic substance to irritate and collapse vein walls; or cyanoacrylate, a surgical sealant (similar to the active ingredient in Crazy Glue), which occludes the vein, preventing blood flow.
Altogether, these therapies constituted a $1.1 billion market in 2018, according to a February 2019 research report from Grand View Research.
Such a large market would seem to be well served, but that’s not necessarily the case, the founders ofVenari Medical found, when they joined the BioInnovate Ireland fellowship in 2016.
BioInnovate is the Irish version of the Stanford Biodesign medical device fellowship, which trains physicians and industry professionals to create medical devices that can go the distance to market and clinical practice. (See “Ireland’s BioInnovate Program: Helping to Build Ireland’s Medtech Hub,” MedTech Strategist, July 29, 2016.)
In 2016, the year that Venari’s founders participated (CEO Stephen Cox, a former computer scientist, CTO Sean Cummins, a biomedical engineer who formerly worked at Cook Medical, and Chief Medical Officer Nigel Phelan, MD, an orthopedic surgeon) the theme was vascular disease. As is customary, the program gave the entrepreneurs access to a network of clinicians during ten months of research to discover clinical problems in need of solutions.
“We found that it was not well understood in the industry that patients suffering from chronic venous disease suffer from significant quality of life impairments.”
The team followed interventional cardiologists, vascular surgeons, and cardiothoracic surgeons in operating rooms, outpatient clinics, and office settings. Cox says “We looked at everything, head to toe, and settled on the venous side. We found that it was not well understood in the industry that patients suffering from chronic venous disease suffer from significant quality of life impairments.”
As their problem definition, the team set out to take the risks out of current treatments and come up with a less invasive and effective approach for patients experiencing more advanced stages of venous insufficiency, in particular, patients already suffering from venous ulcers as a result of their underlying circulation problems.
Venari Medical: A Patient-Friendly Treatment that Doesn’t Involve Burning, Poisoning, or Gluing
Current office-based vein treatments either burn, chemically damage, or glue tissue. Let’s start with thermal ablation, or heating the diseased vessel with laser or RF energy, which constitutes the bulk of treatments (90% of the market). It’s not a procedure that many patients would be willing to undergo a second time, notes Cox, because preparation for the therapy is cumbersome and painful.
Thermal ablation is not precisely targeted, since heat can propagate out from the treatment zone and damage surrounding nerves and tissues. For this reason, prior to the ablation, clinicians inject the patient 10-20 times to infuse anesthesia and high volumes of saline to insulate the surrounding skin and nerves from injury.
“We saw a number of patients in tears while undergoing current market-leading treatments,” says Cox. Not only does that make for a bad patient experience, he says, “it is also time-consuming for patients and clinicians, and they don’t return to get subsequent veins treated so the disease gets worse.” Thermal energy also presents a small risk of DVT, due to forward-firing thermal energy penetrating the vessel at some length.
Sclerotherapy, the next alternative, involves the injection of a substance (usually a chemical detergent) that disrupts the inner vein cellular lining, causing the vein to collapse and shut down over time. This might offer a better patient experience, but it’s less effective than thermal ablation. While thermal ablation succeeds in eliminating the affected vein 90% of the time, efficacy rates of sclerotherapy average 50-80%, and repeat procedures are often required to eliminate symptoms.
Finally, while cyanoacrylate (which is delivered by Medtronic’s VenaSeal vein closure device, for example) is as efficacious as thermal ablation, it remains in the body and can cause a hypersensitivity reaction in some people, necessitating, in some cases, the removal of the affected vein.
For the Venari team, the question became “How can we approach the disease the same way and achieve the same results as best-in-class thermal energy without using burning, chemicals, or glue?”
The company opted for a mechanical approach to vein closure, founding Venari Medical in 2018. With approximately€550,000 in support from Enterprise Ireland and EIT Health, as well as participation in the MedTech Innovator and TMCx, the Texas Medical Center’s start-up accelerator, in two years the company has protected its IP in Europe and the US, completed five preclinical studies to prove the safety and efficacy of the device, and is preparing for first in human trials next year.
A Low-Risk Office-Based Treatment
Venari’s endovenous BioVena therapy will initially target truncal varicosities of the great saphenous vein. The device uses a nitinol shape memory coil to disrupt the inner lining of the vein in order to trigger the body’s natural healing process to close the vessel.
The tip of the flexible device, which is sheathed in a 4 Fr delivery catheter, has a micro- and macro-abrasive surface to achieve the greatest possible degree of cell disruption without tearing or puncturing the vein wall. Using the intuitive handle with one hand and the ultrasound probe on the surface of the body with the other, the operator introduces the catheter into the vein at the knee or ankle. Once in place, using the handle’s thumbwheel, the clinician unfurls the mechanical helical structure until it exerts radial force on the vein wall. Using an intuitive withdrawal motion, the device is pulled back towards the entry site to treat the vein. After treatment, another turn of the thumbwheel re-sheaths the abrasive tip and the catheter is withdrawn from the body.
The most challenging aspect of development, says Cox, was to achieve the proper degree of cell disruption to cause scarring without tearing or perforating the vein wall. “The secret sauce is that you have to do it in a flexible, compliant way, because vein walls are very flexible and thin. You have to disrupt, at a cellular level, without tearing the vein, and you also have to be able to navigate through vein constrictions, such as vein valves, encountered regularly during treatment of large sections.”
Cox notes that the therapy is likely to be free of pain. “It only requires one injection at the access site. And because we are only treating the inside of the vein to a very small depth—a matter of microns deep—we won’t cause pain, because the nerve bundles are in the adventitia on the outside of the vein wall,” Cox says.
Compared to thermal ablation, BioVena also shaves off 20 minutes of procedure time, another competitive advantage for an office-based procedure. “It allows the physician to treat one extra patient every two hours,” Cox says.
The company is currently preparing for a design validation and verification phase next year, in preparation for a first-in-human study mid next year.
Addressing the Greatest Clinical Need
Venari’s market entry strategy revolves around patients with the most advanced disease, where it thinks its therapy should excel. Cox points out that venous leg ulcers, which constitute 80% of all leg ulcers, cost health systems $10,000 per patient, per year. From clinical trials like EVRA, evidence is also emerging to support the notion of treating underlying venous disease early in the course of venous leg ulcers to improve outcomes in terms of faster healing times and lower ulcer recurrence rates.
Cox points out that due to gravity, blood pools at the bottom of the leg, and that’s where venous ulcers occur. “Anatomically, this poses higher risk for thermal ablation because of the proximity of the saphenous and sural nerves. If you try to close the vein by burning, you’ll face a higher risk of skin and nerve injury. That’s where we can add significant value to these venous ulcer patients.”
When asked whether the time has finally come for the venous device market, Cox responds, “The time is now for the patients. Patients with open wounds in their legs that are destroying their lives have seen little innovation in their treatment options over the past 20 years.” There are three million patients in the US and EU with venous ulcers that result in open sores. “They have been largely ignored because superficial venous insufficiency is either seen as a cosmetic issue, or one that has already been solved.”
Because the Venari device is purely mechanical, Cox feels it is uniquely positioned to treat more advanced stages of disease, the venous leg ulcer patients. “These patients are underserved and have the potential to benefit greatly from earlier interventions which BioVena will aim to deliver in a less invasive way than ever before.”
The company is currently preparing for a Series A funding round to support first-in-human studies.
InterVene: A Surgical Predicate Yields a New Endovascular Treatment
InterVene is among a group of companies attempting to address the underlying cause of deep venous reflux by restoring functional vein valves to diseased veins, including InnoVein Inc., which was founded by Albert K. Chin, MD, a serial medtech entrepreneur who goes all the way back to the founding of Guidant Cardiac Surgery; and Hancock Jaffe Laboratories Inc., which went public in an IPO last year.
InterVene decided at the outset to leave nothing behind, by treating veins with an endovascular devices that uses the veins’ own walls to create a vein valve.
Most companies in the space are placing permanent implants in the veins to create valves, but InterVene decided at the outset to leave nothing behind, by treating veins with an endovascular device that uses the veins’ own walls to create a vein valve.
InterVene’s CEO Fletcher Wilson notes that implants in the leg veins face several challenges. First, blood moves slowly in that region, increasing the odds of implant thrombosis and necessitating long-term anticoagulation. And the consequences of implant thrombosis are dire; expensive emergency bailout procedures to remove clots or devices.
Founded in 2011, InterVene spent its early years as a company in residence at the Fogarty Institute, but had its start at the Stanford Biodesign Innovation Fellowship. (See “Biodesign at Fifteen,” MedTech Strategist, June 2016). Fletcher Wilson and his team in the Biodesign class of 2009-10 were focused on unmet needs in vascular disease.
He says they were primarily looking at vascular problems associated with hemodialysis, when they encountered many patients with venous disease. Says Wilson, “I talked to one man in the hallway on the verge of tears, because he had come to Stanford every year for the last eight years and was tired of the fact that even a prestigious institute like Stanford could give him nothing but tight socks,” (i.e. compression stockings).
The Biodesign team knew they had their mission when team member James Yu, MD, a San Francisco Bay-area urologic oncologist uncovered, in the literature, a surgical procedure for creating vein valves out of a patient’s own tissue. The goal became to develop an endovascular version of that very invasive procedure, to create vein valves for deep vein reflux without leaving behind an implant.
Wilson went to Modena, Italy to learn from Oscar Maleti, the surgeon who in 2005 had created the surgical procedure, called Neovalve, to help his patients with venous ulcers. Upon Wilson’s return, the team got to work to mimic some of the surgical results, but with catheter-based tools.
Getting at the Underlying Cause of Venous Reflux
With a focus on deep vein reflux, an area of venous disease not currently served by an endovascular approach, the team developed a catheter-based technique that allows the physician to see what he or she is doing while performing delicate maneuvers to create a new vein valve, a kind of kangaroo pouch on the inside of the vein, and potentially more than one of those in a given procedure.
The BlueLeaf 16 Fr catheter is inserted into the common femoral vein from above, in a retrograde direction. The operator uses intravascular ultrasound to image the vein and help find a location in which to form a valve, basically a clean section of the vein. “We aren’t fixing old valves,” says Wilson, “we are creating new ones.”
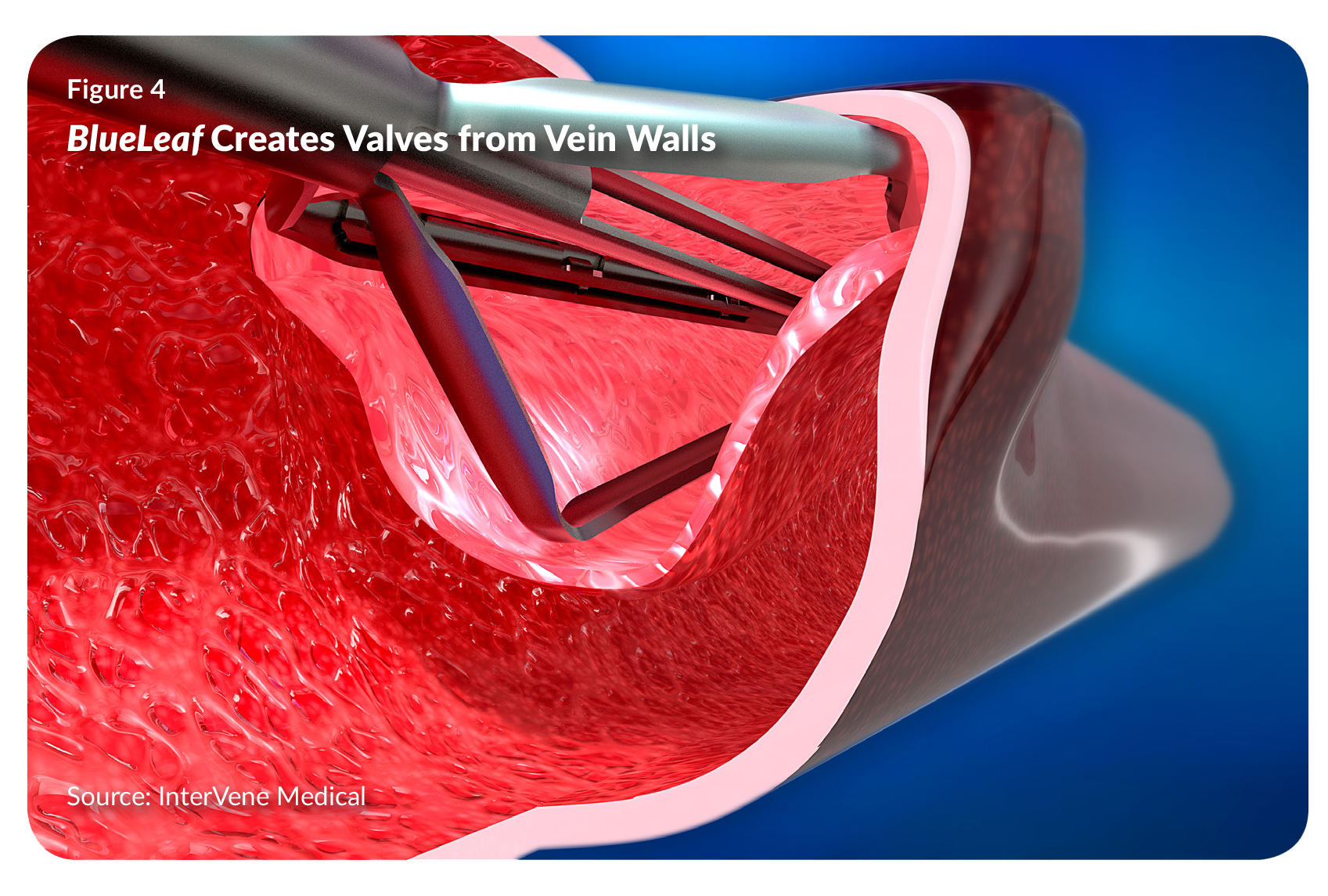 The catheter engages the vein wall by inflating a compliant balloon, which pushes the wall down onto a recessed surface, the distal end of the catheter. That allows the operator to gain access to a layer within the vein wall, using a needle and a special approach called hydrodissection. As the needle pierces into the wall, which is held taut by the catheter, it injects a stream of pressurized saline to create a dissection layer within the wall. “It isn’t cutting; it’s opening up an intramural dissection pocket and access plane,” says Wilson (see Figure 4).
The catheter engages the vein wall by inflating a compliant balloon, which pushes the wall down onto a recessed surface, the distal end of the catheter. That allows the operator to gain access to a layer within the vein wall, using a needle and a special approach called hydrodissection. As the needle pierces into the wall, which is held taut by the catheter, it injects a stream of pressurized saline to create a dissection layer within the wall. “It isn’t cutting; it’s opening up an intramural dissection pocket and access plane,” says Wilson (see Figure 4).
Under intravascular ultrasound guidance, the operator confirms that the needle is within the vein wall, along a length of vein sufficient to form the valve. The operator then passes a dissector into the wall and an actuatable tool (similar to one that surgeons use) opens up a width and length of pocket, and incises the top. It thus separates a leaflet from the wall, completing the kangaroo pouch geometry that mimics, as closely as possible, the physiology of natural vein valves.
InterVene is currently engaged in clinical trials in Australia, New Zealand, and Canada, and has gained approval to do an IDE study in the US, which hasn’t yet begun.
At the moment, InterVene is also focused on building out its team to gear up for pivotal trials. Jeff Elkins recently joined as COO. He is a veteran of both the endovascular industry on the arterial side, having worked at several endovascular start-ups acquired by strategic companies, and the venous space, as the former CEO of Veniti Inc. (which, as noted earlier, is now part of Boston Scientific).
According to Wilson, InterVene’s pivotal trial will be “The first of its kind, so there is a lot of clinical, regulatory, and reimbursement-related work to be done to properly design the study, including building out the team, engaging with the FDA, and payors. We’re doing a lot of that right now.”
To date, the company has raised just shy of $30 million up through the Series B round (which took place in June 2019), from a group of investors that includes RiverVest Venture Partners, 3x5 Partners, Correlation Ventures, Boston Scientific, and angel investors.
InterVene is on track to be the first company to enter pivotal trials with an endovascular treatment that creates new vein valves. In the long run, if competitors developing vein valve implants succeed in proving that they have equal efficacy, InterVene will still have more than a first-mover advantage, notes Wilson. “The ‘ask’ for the patient and the medical community is so much lower if you don’t leave a permanent implant behind, especially since some of these patients are younger. An implant-less approach theoretically has less downside risk, lower long-term requirements for anticoagulation, and ‘re-doability’ over time, if needed.”
Not A Small Problem
InterVene’s Wilson believes that the day for venous devices certainly is coming. “For a while, venous was thought of as a lifestyle problem. But it turns out that there are even more patients here than was previously recognized. It’s not a small problem.”
And, as hospitals scrutinize areas of high cost to see what can be done about them, venous devices have a role to play. “These patients are getting venous ulcers over and over again. They are costing $15 billion in direct costs to the US healthcare system every year, which is about one dollar out of every 200 spent,” says Wilson. “This is a huge problem. I predict that it will be eventually bigger than peripheral vascular disease. There are more patients, and it is a persistent problem.”
Besides, there is a human side to the story, Wilson says. “Another thing that has attracted clinicians to venous over the last five years is how quickly patients get better, especially in the venous stenting world. Patients come in, in terrible shape, with a bad blockage. The next day after the procedure, they’re calling the doctor to say how much better they feel.”

