ARTICLE SUMMARY:
Advanced cardiac imaging will support interventionalists as they perform more complex transcatheter procedures. NuVera’s NuVision™ ICE Catheter aims to play a critical role in shaping the future of 4D ultrasound imaging. Sponsored by NuVera Medical.

NuVera Medical
NuVera Medical, Inc., a portfolio company of Shifamed, LLC., is enabling a new era in transcatheter cardiac interventions through the advancement of real-time 3D intracardiac echocardiography (4D ICE).
Real-time 3D (4D) intracardiac echocardiography (ICE) compared to 2D ICE is analogous to the difference between a black and white television and 3D IMAX movie.
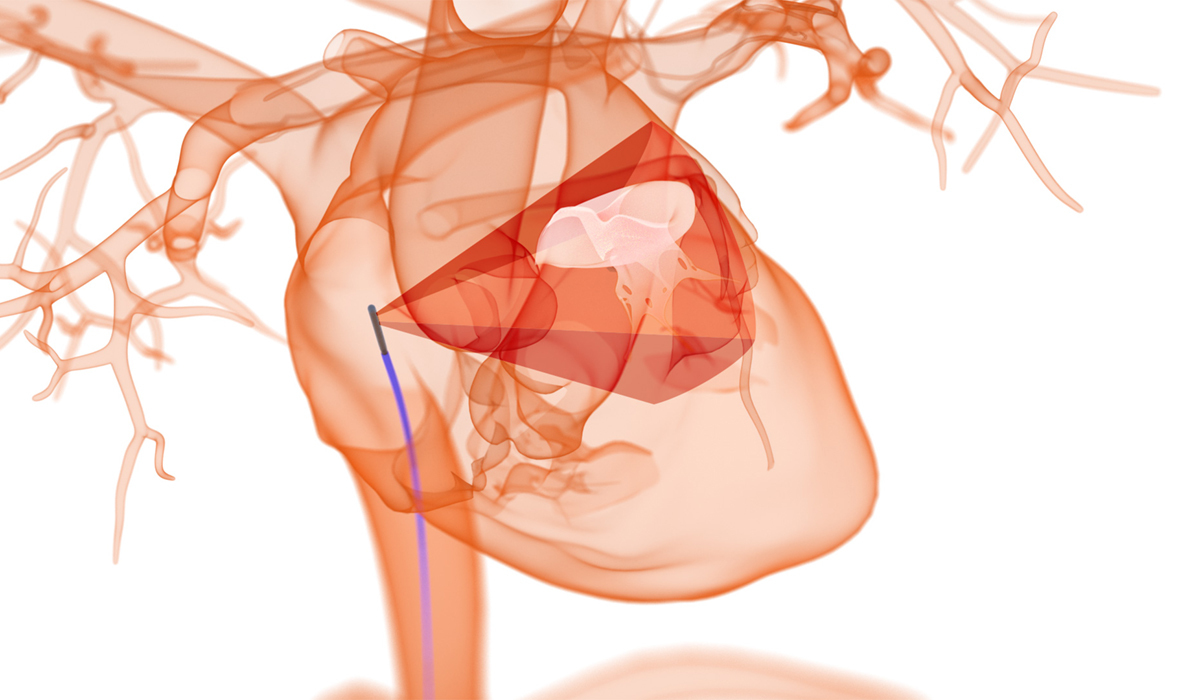
This next-generation, evolving imaging modality combines 3D images with the fourth dimension of time, allowing interventional cardiologists and electrophysiologists to have “eyes on the inside” in real time, a critical component to guiding complex cardiac procedures.
While traditional imaging modalities of 2D and 4D transesophageal echocardiography (TEE) and 2D ICE have successfully guided structural heart and electrophysiology procedures over the past decade, they hinder meaningful therapeutic advances, particularly in the rapidly evolving structural heart market.
Figure 1. Tricuspid Valve Comparison
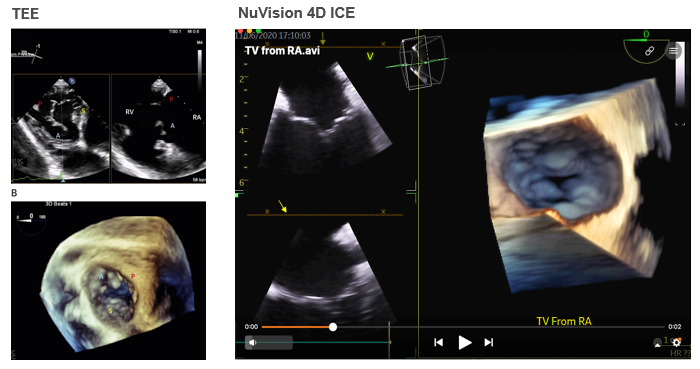
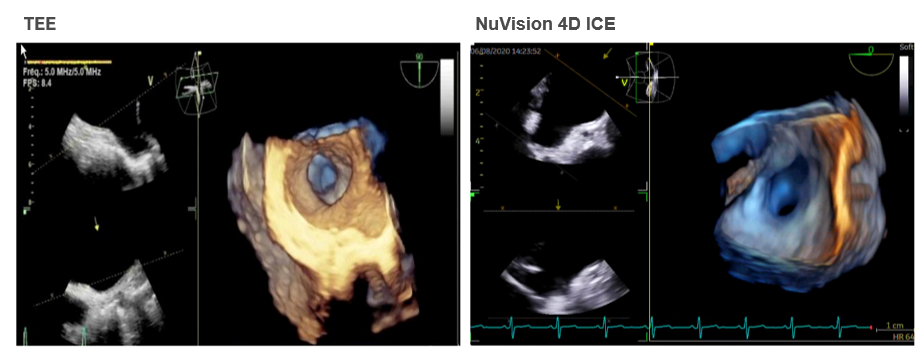
Figure 2. LAA Comparison
Current solutions offer limited views and challenging catheter and probe manipulation, insufficient for guiding delicate procedures in complex cardiac anatomies, such as left atrial appendage (LAA) closure, mitral valve repair, and tricuspid valve repair. Additionally, such procedures must be performed under general anesthesia, requiring dedicated staff, and adding significant time, which increases cost and radiation exposure to the patient and physician.
NuVision™ ICE Catheter: 4D Imaging on an ICE Platform
NuVera Medical, a portfolio company of the highly specialized medical innovation hub Shifamed LLC, is enabling a new era in transcatheter cardiac interventions through the advancement of current 4D ICE imaging. The company’s NuVision™ ICE Catheter is designed to offer a 90°x 90° field of view and high-resolution image rendering in real time. This provides an accurate depiction of cardiac structures and hemodynamic data to facilitate fast intraprocedural decision-making and streamlined workflow.
“With the NuVision ICE Catheter I can view complex intracardiac structures from an entirely new perspective, providing a more accurate picture of cardiac function compared to traditional TEE or 2D ICE,” states Azeem Latib, M.D., Director of Interventional Cardiology and Structural Heart Program Interventions at the Montefiore Medical Center in New York. “4D ICE will unquestionably become a useful modality for the guidance of my structural heart therapies.”
The catheter’s dual-shaft design allows the physician to independently rotate the transducer tip, optimizing its orientation during the procedure, enabling quick and easy visualization from biplane and multiplane views. This vantage point helps physicians assess the complete intracardiac landscape to support more accurate device sizing, evaluate implanted device positioning, and help recognize potential complications.
“It simplifies complex procedures for greater physician and patient benefit,” continues Latib. “The positioning probe is easy to use, manipulate, and rotate – a marked improvement over current imaging which has largely stayed the same for years.”
The rapid processing of the NuVision ICE Catheter is designed to reduce procedural and fluoroscopy times and decrease overall radiation exposure to both the patient and physician. Furthermore, the catheter’s femoral access allows the physician to avoid intubation, potentially enabling a better patient experience under conscious sedation, supporting earlier hospital discharge, and reducing costs.
The NuVision ICE Catheter is designed to have a minimal learning curve and to work with conventional ultrasound systems, fitting comfortably into the current workflow, simplifying the transition to 4D cardiac imaging.
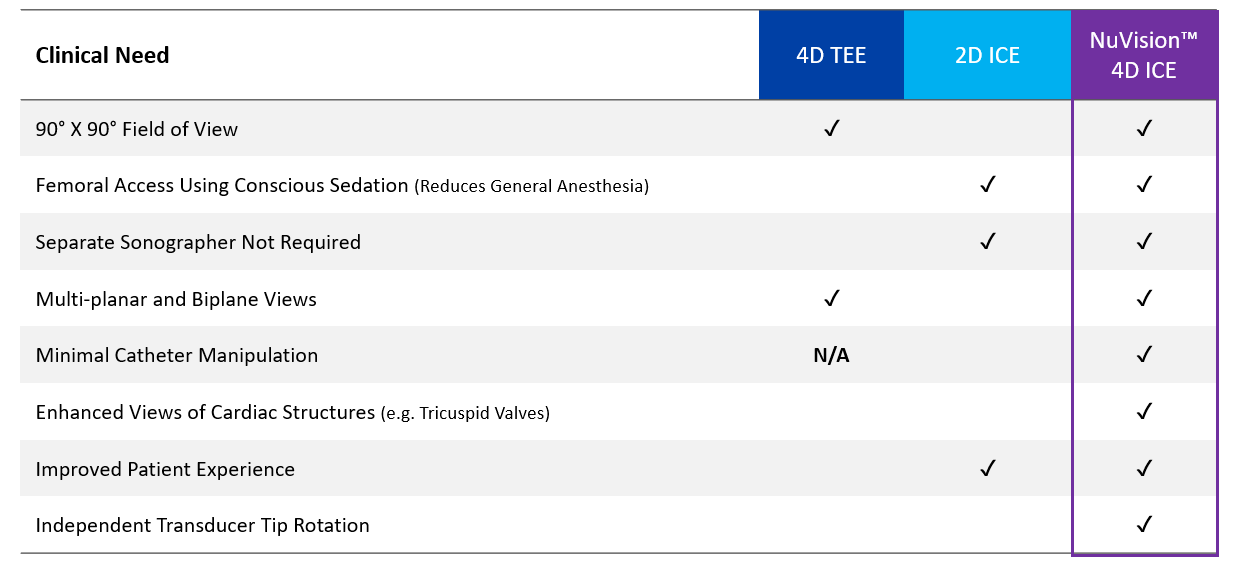
Figure 3. Echocardiography Platforms
Innovators in Minimally Invasive Technologies
NuVera’s NuVision ICE Catheter is crafted from the proven playbook of serial entrepreneur and Shifamed founder, Amr Salahieh. Shifamed has grown to a team of over 150 medtech experts across its multiple portfolio companies and is focused on developing minimally invasive technologies that target high-impact clinical areas for greatest human benefit. In addition to cardiac imaging, current therapeutic areas of focus include high-risk coronary intervention and cardiogenic shock, mitral and tricuspid valve replacement, heart failure, cataracts, presbyopia, and glaucoma.
“The NuVision ICE Catheter represents an entirely new phase of transcatheter interventions, an area formerly limited by general anesthesia requirements and two-dimensional views,” stated Amr Salahieh, Founder and CEO of Shifamed. “NuVera will enable physicians to do much more with their procedures, a potential game-changer for the electrophysiology ablation and structural heart markets.”
Shifamed’s proven development model and infrastructure accelerates the advancement of meaningful, leapfrog technologies. This is backed by the incubator’s decade-long track record of fruitful exits, including the sale of Apama Medical to Boston Scientific in 2017, Kalila Medical to Abbott/Terumo in 2016, Maya Medical to Covidien in 2012, Sadra Medical to Boston Scientific in 2010, and Embolic Protection to Boston Scientific in 2001.
Early Clinical Results
NuVera recently completed its first-in-human study and is mid-way through an initial clinical evaluation of the NuVision ICE Catheter. A total of 33 patients have been studied to date with positive outcomes
First-in-Human Results
The company’s first-in-human feasibility study, conducted at the Italian Hospital Asuncion Paraguay, was used to provide image guidance in the treatment of five patients for atrial septal defects while under conscious sedation. All primary and secondary safety and performance endpoints were met with no procedure or device related adverse events.
Figure 4. ASD Closure (Amplatzer) Procedure
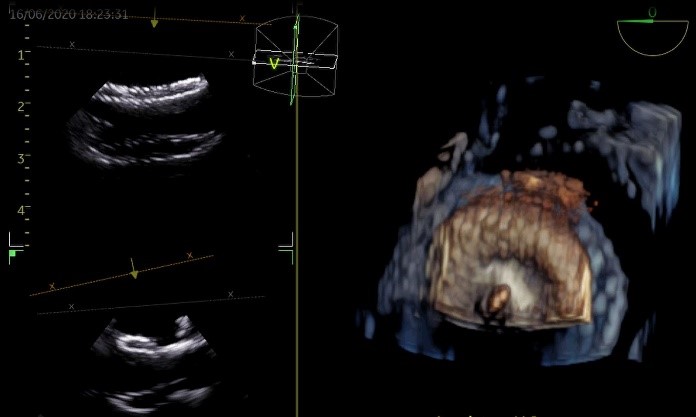
Imaged using NuVision ICE Catheter. Picture Courtesy of Adrian Ebner, M.D., Head of the Cardiovascular Department at the Italian Hospital Asuncion Paraguay
The study was conducted using remote, live-feed support from a team located in the U.S. and was one of the first virtually supported clinical studies to be performed during the world COVID-19 pandemic.
Clinical results of the FIH study were presented at the Transcatheter Cardiovascular Therapeutics (TCT) Connect 2020 virtual conference by principal investigator Adrian Ebner, M.D., Head of the Cardiovascular Department at the Italian Hospital Asuncion Paraguay and Azeem Latib, M.D., Director, of Interventional Cardiology and Structural Heart Program Interventions at the Montefiore Medical Center in New York.
Initial Clinical Evaluation
The NuVision ICE Catheter is also being evaluated in a larger European prospective study of 28 patients across two European sites. Treatment included a variety of structural heart and electrophysiology ablation procedures with many of the patients treated under conscious sedation and without need for fluoroscopy. Treatments included placement of LAA occluders (LAAO), supraventricular tachycardia and premature ventricular contraction, pulmonary vein isolation, ventricular tachycardia, and other complex cardiac procedures.
Primary and secondary safety endpoints were met in all treated patients with no device-related events. All primary performance endpoints were met with operators rating NuVision’s 2D ICE imaging and steering capability superior to currently available 2D ICE catheters with no equivalent comparator to 3D.
“These clinical results are very positive, supporting a novel approach to visualizing the right and left heart, allowing us to treat patients in the same or less amount of time,” stated Amin Al-Ahmad, M.D., cardiac electrophysiologist at the Texas Cardiac Arrhythmia Institute at St. David’s Medical Center in Austin, Texas. Dr. Al-Ahmad participated in the European study.
Figure 5.Left Atrial Appendage
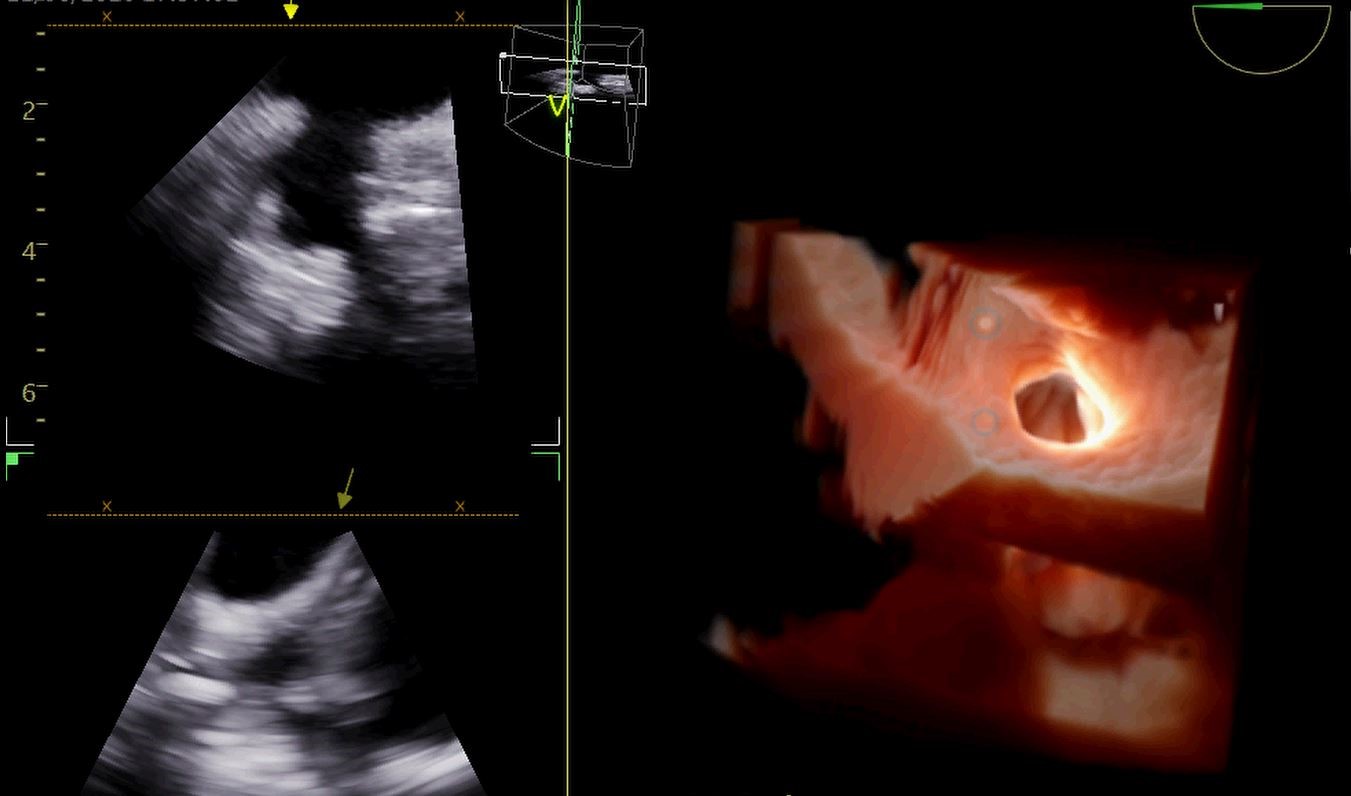
Imaged using NuVision ICE Catheter. Picture Courtesy of Adrian Ebner, M.D., Head of the Cardiovascular Department at the Italian Hospital Asuncion Paraguay
“I am very excited to be studying 4D imaging for use in ablation procedures, which have become increasingly complex, requiring a greater field of view to safely perform,” stated Andrea Natale, M.D., F.H.R.S., F.A.C.C., F.E.S.C., cardiac electrophysiologist and executive medical director of the Texas Cardiac Arrhythmia Institute. “NuVision ICE offers the widest field of view available, is easy to navigate, and has demonstrated excellent safety and performance in its initial clinical trials. I look forward to evaluating it next year when it comes to the U.S.”
Looking Ahead
As NuVera looks ahead to future commercialization, the team will focus on building a strong foundation to drive product adoption initially in the U.S. “We are planning for a limited market release in key U.S. heart centers to focus on promoting product awareness while capturing early market feedback,” stated Anthony Medigo, Chief Commercial Officer at NuVera Medical. “We will further strengthen our physician relationships with plans to establish centers of excellence as part of our robust clinical program strategy.”
“We are extremely pleased by the early performance and positive physician response to the NuVision ICE Catheter,” stated Todor Jeliaskov, CEO and President of NuVera Medical. “I am eager to achieve the next major milestone of U.S. regulatory clearance and bring this beneficial technology to market in the very near future.”
Figure 6. Tricuspid Valve
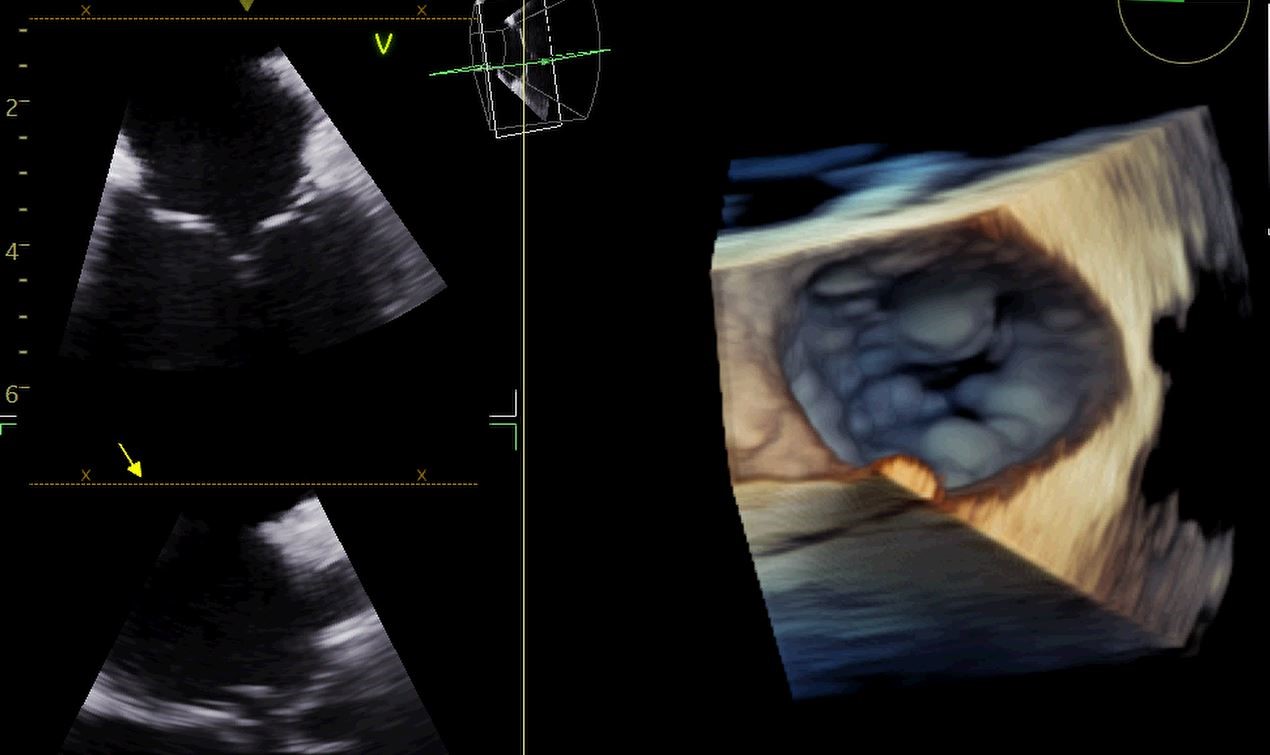
Imaged using NuVision ICE Catheter. Picture Courtesy of Adrian Ebner, M.D.
All figures courtesy of NuVera unless otherwise noted.
About NuveraNuVera Medical, Los Gatos, CA CONTACT: Todor Jeliaskov, CEO FOUNDED: 2016 WHO IS BEHIND IT: Amr Salahieh, founder of the highly specialized medical innovation hub, Shifamed LLC and its team of 150+ development and production engineers, finance, marketing, clinical and regulatory experts. Todor Jeliaskov, President and CEO, previous GM and VP of Global Ultrasound at GE Healthcare. Anthony Medigo, Chief Commercial Officer, former VP of Interventional Therapies at Siemens Healthineers. UNMET CLINICAL NEED: Challenges in procedural guidance, underserved by the capabilities of 2D ICE and standard TEE, hinder therapeutic advances in electrophysiology ablation and rapidly evolving structural heart interventions. SOLUTION: The NuVision™ ICE Catheter is designed to offer high resolution 4D ultrasound imaging, allowing physicians to uniquely visualize complex interventions. This provides for a more efficient workflow, potential to eliminate general anesthesia, reduce radiation exposure, and improve costs. FUNDING TO DATE: NuVera recently extended its Series B round of funding, a convertible note, from $15 million to $19.8 million, following an earlier $11 million equity round in Series A. |
NuVera Medical, Inc., a portfolio company of Shifamed, LLC., is enabling a new era in transcatheter cardiac interventions through the advancement of real-time 3D intracardiac echocardiography (4D ICE).
Learn more at NuVera Medical